Tungsten Trioxide: A Comprehensive Commentary
Historical Development
People discovered tungsten trioxide (WO₃) while searching for new ways to color ceramics and glass. Chemists in the late 18th century were taken with heavy mineral ores like scheelite and wolframite. They noticed bright yellow residues after treating these ores with strong acids. By the early 1800s, scientists figured out this yellow stuff acted as a strong oxidizer and that heating it could release metallic tungsten. The groundwork laid by those early European chemists still shapes how the material gets used in the present, especially in electrical and chemical industries. The road from laboratory oddity to industrial workhorse involved repeated refinement, driven by demand for brighter, longer-lived pigments and glow-in-the-dark lighting.
Product Overview
Tungsten trioxide usually appears as a lemon-yellow, fine powder. It acts as a strong oxidizing agent and packs a heavy punch, both in terms of density and reactivity. Its crystalline structure makes it tough and stable, so it resists breaking down unless exposed to high temperatures or strong reducing agents. Manufacturers favor this compound because it easily morphs into different forms—thin films for electronics, layered structures for solar devices, or dense bodies for wear-resistant coatings. Found on lab benches and shop floors alike, tungsten trioxide serves a variety of needs, providing options for both large- and small-scale users.
Physical & Chemical Properties
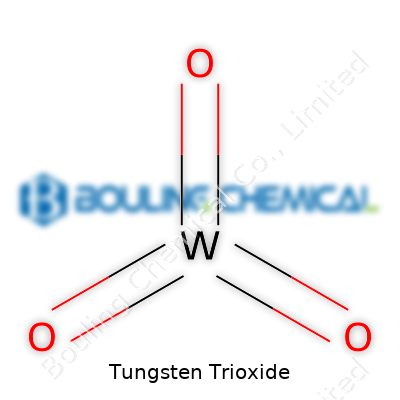
WO₃ stands out for its vivid yellow color, high density (about 7.16 g/cm³), and melting point in excess of 1400°C. It feels gritty between your fingers and resists easy dissolution in water, yet it comes alive in alkaline solutions thanks to its amphoteric character. This chemical switches between different oxidation states, giving it flexibility in electric and optical devices—one reason for its starring role in smart windows and sensors. Its powdery consistency can create dust, warranting decent ventilation during handling. As a crystalline oxide, its particles show sharp edges under the microscope, with a tendency to clump without additives.
Technical Specifications & Labeling
Bags and barrels of tungsten trioxide get labeled with purity percentages, trace-metal contents, particle sizes, and moisture levels. Grades vary widely, depending on whether the powder feeds into pigment production, hard metals, or thin-film electronics. High-purity options, reaching up to 99.999%, command higher prices. Manufacturers check for contamination from other transition metals, which can throw off electrical or optical performance. Customers often demand certificates of analysis that list not only the basic chemical content but also the residual acids and halides. Packing must shield the contents from moisture or acids, since both degrade purity.
Preparation Method
Most industrial sources of tungsten trioxide start with minerals like scheelite or wolframite. Miners crush and grind those rocks, extract the tungsten using sodium carbonate, and then filter out the sodium tungstate solution. Chemists acidify this solution, which causes yellow WO₃ to drop out. After careful washing and drying, the powder meets strict industrial requirements. Smaller labs may roast tungsten metal in oxygen at high temperature, but this route costs more and scales poorly. Both paths aim to avoid metallic or non-metallic impurities, as these wreck downstream processes or weaken final products. A personal trial with laboratory-scale synthesis showed that keeping acids pure made a day-and-night difference in the brightness of the final yellow product.
Chemical Reactions & Modifications
WO₃ stands ready to swap oxygen atoms, especially in the presence of hydrogen or carbon monoxide, thanks to its strong oxidizing ability. Engineers exploit this for catalytic reactions—hydrogenating organic compounds or scrubbing pollutants from exhaust streams. Electrochemists customize its surface by injecting ions like lithium, creating “tungsten bronzes” that shift color or conduct electricity more readily. Experienced glassmakers and ceramists learned long ago how slight tweaks in the formula—such as a touch of sodium—change how the oxide melts and bonds, giving better frit or glaze performance. Classic textbooks detail how WO₃ plays well with acids and bases, forming tungstates or dissolving in the right alkaline bath. My own tinkerings with molybdenum analogues drove home how subtle those differences in tungsten chemistry really matter for films and coatings.
Synonyms & Product Names
On the shelf or invoice, tungsten trioxide might appear under trade names like “tungstic anhydride” or just “tungstic oxide.” In old literature, you can still find “yellow oxide of tungsten” or, rarely, “wolframic anhydride.” The standard chemical abbreviation is WO₃, but forms like ammonium paratungstate and sodium tungstate crop up in precursor or derivative work. Global suppliers may list the product using CAS Number 1314-35-8 to avoid confusion. The variety of names and languages across product catalogs can cause headaches for procurement staff, reminding buyers to always check technical datasheets rather than just the label.
Safety & Operational Standards
Material safety data files for WO₃ warn about inhaling dust—this heavy powder may settle in the lungs if handled without control. Gloves and goggles keep fine particles off skin and eyes, especially when pouring from bulk drums. Workers run fume hoods or dust extractors, since long-term respiratory exposure raises concerns, even if major studies show tungsten trioxide causes few acute toxic effects. Environmental standards look for proper disposal, since this compound resists breakdown in soil and water. Regulations sometimes require secure containment, since small spills can spread widely—making cleanup a hassle. My own shop always keeps vacuum cleaners with HEPA filters nearby, since a broom just stirs up even more powder.
Application Area
Industries lean on tungsten trioxide for diverse reasons. As a pigment, it colors ceramics, tiles, and glass with a persistent yellow that never fades. Metallurgists use it as an intermediate, turning WO₃ into metallic tungsten for lightbulb filaments and heavy-duty electrodes. The electronics trade values WO₃ for smart-glass windows and low-voltage sensors, where its capacity to switch oxidation states controls light or electricity. Environmental engineers tap it for pollution control—both as a catalyst and in visible-light photocatalysis. It also appears in x-ray shielding materials, pigmenting rubber, and even as a neat additive in certain steels for aerospace. I’ve seen WO₃-based products spread across research labs, commercial greenhouses, and art studios alike.
Research & Development
Scientists keep hunting for new twists on this well-established oxide. Research centers today focus on two main tracks: energy storage devices and environmental catalysts. Battery makers test thin films of WO₃ as electrodes for next-generation lithium-ion batteries. Solar panel designers push for higher performance using WO₃’s unique photochromic properties. A friend at a university worked with doped tungsten trioxide for gas sensors, showing how changing the mix with other metals can improve detection limits for pollutants. Meanwhile, teams explore how nanostructured WO₃ spins up faster reactions, aiming for greener chemical processes or improved carbon capture. It seems almost every year brings a new patent filing, keeping tungsten trioxide squarely in the crosshairs of innovation.
Toxicity Research
Toxicologists have scrutinized tungsten trioxide for decades, and the results provide some comfort. Studies show it passes through the body rapidly and doesn’t accumulate in organs the way some heavy metals do. Animal tests at low to moderate doses point to minimal harmful effects, and workplace cases of severe poisoning are rare. Still, no one ignores the hazards of chronic dust inhalation. Regulatory agencies call for rigorous industrial hygiene to limit airborne concentrations. Medical researchers remain vigilant, especially for long-duration or high-dose exposures, which might carry unknown long-term risks. Laboratory animals exposed to high concentrations sometimes show mild inflammation, but not severe lung disease or cancer. That said, a few open questions remain about environmental persistence and ecosystem disruption—reminding operators to handle the powder with the same respect they give other transition metal oxides.
Future Prospects
Looking ahead, demand for tungsten trioxide seems poised to grow. Smart windows and optoelectronic devices reach deeper into everyday infrastructure, and the distinctive electronic and optical features of WO₃ stand out as a solution candidate. Energy researchers eye this oxide for tougher, longer-lived batteries and capacitors, buoyed by its chemical resilience. Pollution-control technologies scan WO₃ for roles in large-scale catalytic converters, especially as environmental standards tighten worldwide. As more research uncovers new doped versions or engineered nanostructures, the potential market continues to spread. Economic shifts and surplus supplies sometimes rattle the price, but the track record for tungsten trioxide suggests enduring relevance across many sectors. In my own work, the shift from bulk powders to carefully engineered nano- or thin-film materials points toward a future where WO₃ gets tailored for ever more demanding and creative applications.
Why Tungsten Trioxide Matters
Tungsten trioxide shows up in more places than most folks realize. In my work helping people find solutions for energy and sustainability, the uses for this bright yellow compound crop up again and again. Power plants, tech companies, and even artists find value in this strange but useful powder. Its unique characteristics, like toughness under stress and the way it interacts with light and electricity, create many real-world opportunities.
A Helping Hand in Clean Energy
Energy storage stands out as one of the most important areas for tungsten trioxide. Companies working on batteries often look for materials that last long and handle plenty of charge cycles. Tungsten trioxide has delivered in new types of lithium-ion and sodium-ion batteries. Thanks to its stable structure, it puts up with the expansion and contraction that wreaks havoc on other battery ingredients. There’s real progress in using it to make cheaper and more durable grid-scale storage possible, which helps give a leg up to wind and solar projects. Serious scientific reviews back up these claims, and a quick look at recent research from respected labs (Nature Communications, 2021) shows that tungsten trioxide often outperforms older materials, especially under tough conditions.
Making Smart Windows Smarter
Buildings burn a lot of energy keeping spaces comfortable. Smart glass uses tungsten trioxide for dynamic tinting—think windows that get darker on sunny days or clear up when the clouds roll in. With just a low electric charge, these coatings shift how much light and infrared heat travel through. More efficient buildings put less pressure on the power grid and leave people with lower electricity bills. It’s not just about science fiction architecture; today, real office towers in cities like Toronto and Shanghai already rely on this kind of technology. Smart glass also improves privacy and makes living spaces feel more welcoming.
Safer X-Rays and MRI Environments
Hospitals and clinics always hunt for safer ways to shield both patients and equipment from stray radiation. Tungsten trioxide, with its high density, plays a crucial role in coats and screens that stop X-rays and gamma rays in their tracks. In the past, lead filled those jobs but created hefty health and environmental headaches. Over 20 years, regulations around medical waste and staff safety have gotten tighter, which pushes healthcare designers to swap out lead for alternatives. Current evidence from the FDA and medical device industry shows tungsten trioxide remains one of the best candidates, without the toxic legacy.
Color and Art in Industry
Artists and manufacturers still use tungsten trioxide to create vibrant yellow colors in ceramics and glassware. Its ability to hold color even at high firing temperatures brings out sharp details and bold tones in pottery found in homes and museums. Some of the finest glass art relies on that specific hue, and even car factories use these colorants for dashboard indicators and warning lights. For something born in a lab, tungsten trioxide brings a human touch to daily objects.
Not Just for Scientists
From saving energy in buildings, storing the power from wind farms, helping out at hospitals, or even brightening up a piece of pottery, tungsten trioxide forms part of stories most people never hear about. Its job may look simple, but its impact spills over into our comfort, safety, and even beauty. As more research comes out and the push for greener technology keeps growing, the uses for this unassuming yellow compound will only spread further. Companies and communities willing to test out new ideas will continue to find surprising value in something once limited to science textbooks.
Understanding Tungsten Trioxide Risks
Tungsten trioxide, a yellowish powder used in electronics, ceramics, and smart window glass, isn't a household name. But, just like any industrial material, it raises questions about safety once people start handling it in real environments. Stories circulate in lab corridors and factory floors about odd symptoms or sudden rules about protective gear. Sometimes, such warnings spark more anxiety than answers, especially if official reports sound vague or simply cite guidelines without context.
Looking into the Real Hazards
A lot of materials in manufacturing have some downsides if handled the wrong way. Most people never touch tungsten trioxide, but workers in factories or researchers in labs come in contact with it daily. Dust from powders like tungsten trioxide can end up airborne, raising the risk of breathing it in. Inhaling any fine particulate matter, not just chemicals, often leads to coughing, throat irritation, or worse if exposure continues. The Material Safety Data Sheets (MSDS) for tungsten trioxide flag respiratory irritation and sometimes skin and eye irritation, but they often stress that the compound isn't outright poisonous in the traditional sense.
The difference between dangerous and “toxic” can be confusing. Lab tests show tungsten trioxide has low acute toxicity, meaning a one-time exposure doesn’t generally lead to severe effects. Chronic exposure, though, brings up other issues. While there’s no strong evidence that tungsten trioxide causes cancer, long-term inhalation of dust can put stress on the lungs. Some animal studies link tungsten compounds to organ changes after long exposure. Still, health agencies like OSHA and NIOSH haven’t labeled it a carcinogen or set strict personal exposure limits.
Worker Experience Fills the Gaps
Health and safety classes rarely talk about tungsten trioxide but regularly drill the value of dust masks, gloves, and eye protection. During my own time working in a materials research lab, I wore basic safety gear when weighing and mixing tungsten trioxide. Even for decades-long workers, routine health checks didn't show any obvious problems linked to the compound. Still, friends complained about itchy skin and watery eyes if powder landed on uncovered arms or near their face. Ventilation systems made a clear difference; stronger airflow or proper fume hoods meant fewer complaints.
One doesn’t need to be a chemist to see why some caution makes sense. Fine yellow dust settles everywhere, clings to clothes, and hides in tiny crevices. I saw people develop a sense of healthy paranoia, always washing hands before meals and never touching their eyes at the bench. Signs above laboratory sinks reminded workers of tungsten dust’s stubbornness.
Ways to Handle Tungsten Trioxide Responsibly
Safety comes down to informed habits, not just rules on a poster. Managers who invest in good extraction systems and provide easy access to wash stations cut back on unnecessary exposure. Simple steps count—storing powders properly, cleaning workspaces every shift, and reviewing procedures when new folks join the team. Routine air monitoring helps, too, catching problems before anyone feels symptoms. For industries expanding tungsten trioxide use—think smart glass or novel electronics—it becomes especially important to adopt regular training and feedback from workers. Listening to those handling the powder daily often points to risks missed during official reviews.
Tungsten trioxide won’t bring immediate disaster. Yet, treating it with respect follows the best practices for any workplace chemical. With straightforward safety upgrades and honest conversations about health, companies avoid turning a manageable risk into a major headache.
The Story Behind WO3
Tungsten trioxide carries a chemical formula of WO3. At a glance, that string of letters and numbers looks simple enough, but this oxide serves as a hub for one of the less-talked-about branches of modern technology. I always found it a bit surprising how something with such a niche role in textbooks could reach into areas that affect everything from environmental science to the screens in our pockets.
Real-World Impact
Look at the glass on office buildings or the coatings on smart windows. The tiny yellow powder that tungsten trioxide forms isn’t meant for goofy science class experiments — it lets engineers change how much light or heat a window lets through. That gets personal every summer, when the office AC keeps clicking on thanks to a cloudless sky. Scientists figured out how to sandwich WO3 layers into glass to literally dial down the sunlight, not just for comfort but for energy savings. Reports from the U.S. Department of Energy confirm that these sorts of windows cut large building cooling costs by up to 25%.
Manufacturers of LEDs and sensor tech depend on tungsten trioxide too. Its high melting point, excellent chemical stability, and unique way of conducting current in thin films land it in lots of cutting-edge devices. As someone who has worked with electronics recycling, I’ve seen how hard it can get to replace legacy parts unless you know which formula is making a component behave the way it does. WO3 stabilizes colors in certain displays, and acts as a sensitive layer that can sniff out trace amounts of harmful gases. This property is why researchers have flagged it as crucial for next-gen environmental technology.
Health, Safety, and Considerations
Tungsten trioxide does not set off alarm bells for toxicity like lead or cadmium, but any fine industrial dust can affect human health if mishandled. Routine handling of WO3 in a lab setting — say, while preparing a gas sensor — calls for careful ventilation and personal protection, something I learned quickly during my stint maintaining air-monitoring networks. There are currently no widespread, severe health risks linked to tungsten trioxide, according to the European Chemicals Agency and the U.S. Occupational Safety and Health Administration.
Challenges and Where We Go From Here
Extracting pure tungsten to produce WO3 means digging into ores like scheelite and wolframite. Mining brings real environmental scars: water use, habitat loss, leftover rock piles. Circular economy efforts exist, but not nearly on the scale needed to offset today’s demand from electronics, defense, and energy sectors. Reports from the International Tungsten Industry Association show only a fraction of tungsten comes from recycled sources right now.
There’s a growing push for labs and industry to close the loop — reclaiming tungsten from used electronics, adopting greener synthesis, and pushing for transparent supply chains. Supporting recycling programs and researching eco-friendlier chemical processes will decide how widely we keep seeing tungsten trioxide in the future. Its formula might look straightforward, but its story is deeply tangled with how we manage technology, labor, and the environment.
References
- U.S. Department of Energy: Smart windows and energy savings.
- International Tungsten Industry Association: Tungsten recycling statistics.
- European Chemicals Agency: Tungsten trioxide safety profile.
Why Care About Storage and Handling?
Tungsten trioxide comes up a lot in industries tied to electronics, ceramics, and smart windows. Its powdery yellow form can look harmless on the shelf, but looks deceive. Safe handling and smart storage set the difference between routine work and a trip to the emergency room. If a material can mess with your lungs or spark a chemical reaction you didn’t plan for, it asks for respect. I’ve spent enough time in labs with hazard signs taped everywhere. You can’t let your guard down just because you’re used to the routine.
Risks to Workers and the Workspace
The dust from tungsten trioxide irritates eyes and breathing passages. You get this stuff on your skin or in your lungs, it can cause trouble that lingers. I remember a colleague dealt with a week of coughing fits after thinking dust masks were optional. Turns out, tungsten trioxide isn’t considered highly toxic, but that shouldn’t fool anyone. Any fine, heavy metal oxide isn’t worth gambling with—lungs don’t grow back.
Tools and Habits That Work
I grew up hearing, “Measure twice, cut once.” In the lab, this means planning where and how a chemical lives. Tungsten trioxide stays safest in solid, airtight containers. Tough plastic or glass does the job, but you don’t want labels peeling off or lids half-shut. Humidity can clump the powder, and a spill makes cleanup a headache. Keep it in a dry, cool spot that stays away from the main flow of people or places where drinks and food show up. Never mix up your work and lunch areas. I’ve seen real damage from someone grabbing the wrong coffee mug.
Anyone moving or measuring this powder needs gloves, safety glasses, and a lab coat—no exceptions. Some powder always escapes, so a dust mask or, even better, a respirator helps out. Ventilation matters more than most people realize. Fume hoods or local exhaust fans pull dust out of the air before anyone’s lungs do. I’ve seen labs regretting shortcuts on ventilation after a couple of quiet coughing fits spread across the crew. Good routines prevent bad stories.
Disposal Isn't an Afterthought
Tungsten compounds do not belong in the regular trash. Waste routes through marked, sealed containers, and disposal follows hazardous material guidelines that local authorities and environmental agencies set. Skipping proper disposal can lead to soil and water contamination. In my experience, teams get lazy here, especially near shift change or when the workload spikes. Cutting corners on disposal always bites back—sometimes with fines, sometimes worse.
Training Is Real Safety Gear
No one starts a new job thinking they’ll get hurt, but accidents look for complacency. New hires and seasoned pros alike need recurring training, especially as procedures or product lines shift. Straight, hands-on practice with spills, cleanups, and handling beats stacks of paperwork nobody reads. I've seen teams run mock drills, and while it feels silly at first, muscle memory makes a real difference when the pressure's on.
Simple Steps, Big Difference
Keeping tungsten trioxide safe comes down to a few core habits: airtight containers, proper labeling, strong ventilation, gloves, goggles, and responsible disposal. The science is well-known, but the real challenge comes in sticking to these steps every day, even when things get busy or routines get old. You build a trustworthy workspace not with high-tech gadgets, but with steady, everyday habits and clear communication.
Understanding Tungsten Trioxide
Tungsten trioxide, known as WO3 in chemical shorthand, often grabs attention in my conversations with chemists because it stands apart for its unique set of features. It shows up as a bright yellow crystal, sometimes sliding towards a greenish tint if the temperature climbs. You see this yellow powder, and it feels surprisingly dense between the fingertips—much heavier than many other powdered compounds from the same shelf.
Temperature and Color Shifts
This compound’s color doesn’t just provide a visual cue; it signals changes inside its crystal structure. Make it hot enough—several hundred degrees Celsius—and WO3 starts to shift, deepening in color. In research labs, that change allows scientists to track temperature by eye. Not many materials offer this honest feedback, making tungsten trioxide dependable where simple indicators matter.
Density and Structure
WO3 packs plenty of weight for its volume. Its density sits around 7.16 grams per cubic centimeter. For comparison, water sits at only 1, so this powder feels more like finely crushed stone. That weight means it doesn’t drift around the air easily, so even in my experience handling it, cleanup proves far easier compared to fluffy or electrostatically charged powders.
Crystal structure directly shapes how tungstic acid—an old mining term for tungsten trioxide—performs. Its symmetrically packed tungsten and oxygen atoms create a lattice that feels almost stubborn in its durability. You can push, pull, and heat it without warping the basic form, which brings confidence for those putting it through battery testing or high-temperature coatings.
Solubility and Reactivity
Tungsten trioxide doesn’t dissolve in water under standard conditions, so it resists weathering and accidental leaching. Drop some in an acid bath or expose it to a strong base and things change—now it reacts, breaking down in ways useful for catalysts or pigments. This stubbornness in neutral conditions and openness to shift in harsh ones form the backbone of tungsten trioxide’s importance in several industries.
Electrical and Optical Features
WO3 doesn’t conduct electricity well at room temperature, which makes it safer to handle than many metallic compounds. Once voltage or light comes into play, it starts acting a bit differently—changing color or transparency in a process called electrochromism. That’s a big reason why you’ll find it inside smart windows and some rechargeable battery designs. Engineers appreciate this “change on demand” quality, since it allows for smoke sensors and other environmental controls that cycle again and again without losing effectiveness.
Building a Safer Workplace
Handling tungsten trioxide on the factory floor or in a research setting brings its own habits. Its fine particle form urges us to wear masks and gloves—breathing the dust over years can cause irritation. Too many folks ignore this, thinking dense powders don’t reach the lungs. Simple protective gear and good ventilation keep workers healthy and ensure the material stays put until used.
Moving Forward With Responsibility
WO3 holds steady as a solid, heat-resistant, non-flammable powder with a strong personality. Industries rely on these physical properties—distinct color, dependable density, stability, and reactivity in clearly defined circumstances. Mixed with smarter workplace habits, tungsten trioxide continues delivering the features needed for safer chemical processing, energy-saving solutions, and environmental tech.