Triphenylbismuth: In-Depth Insights Into an Organobismuth Compound
Historical Development
Triphenylbismuth crept into the annals of organometallic chemistry at a period when scientists, especially in the late 19th and early 20th centuries, sought to stretch their minds beyond the boundaries of classical metal chemistry. August Kekulé and his followers were among the first to see the promise in aromatic ring structures. Bismuth, heavier and less reactive than its lighter group neighbors, found itself in a unique spot—rarely the subject of as many experiments as carbon or tin. Chemists began preparing triphenylbismuth by reacting bismuth salts with Grignard reagents, establishing a robust path by the 1920s that changed how organometallic molecules could be used. The popularity of triphenylbismuth rose in response to its stability and relatively low toxicity compared to many other heavy metal organics. I’ve watched how research groups would cite old German literature just to trace this compound’s forgotten applications and preparation tweaks, connecting modern labs with chemical pioneers of a hundred years ago.
Product Overview
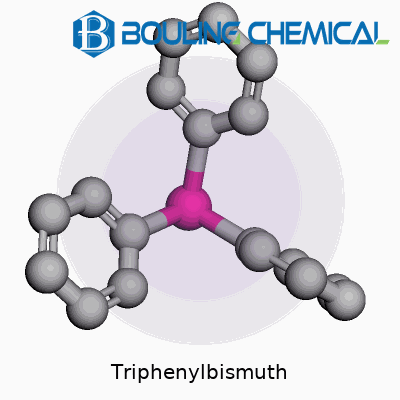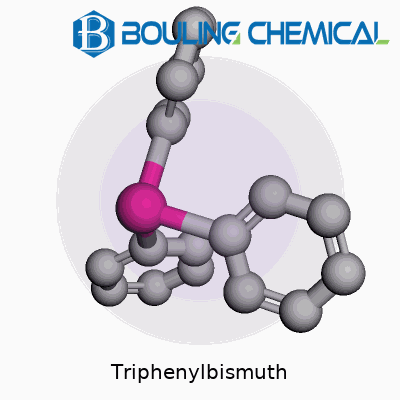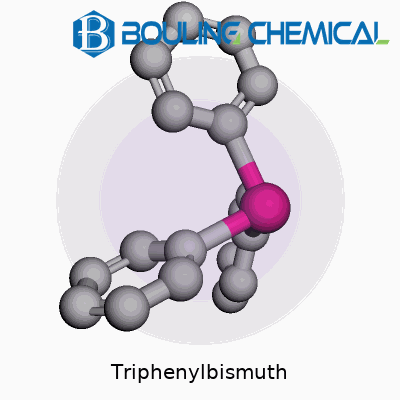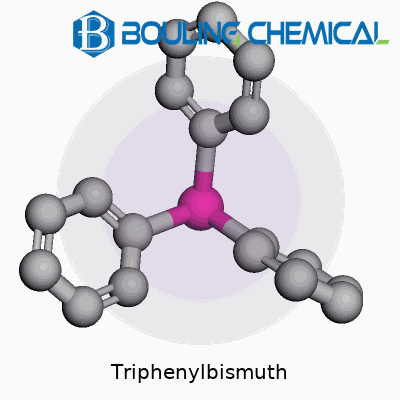
Triphenylbismuth stands out from the crowd of organometallics, packing a bismuth atom bound to three phenyl groups. This pale yellow solid doesn’t crumble under air, making it more user-friendly than many of its peers. Suppliers around the world stock it, and I’ve often found procurement straightforward for both industry-scale and academic research. This compound keeps emerging in diverse applications, acting in roles from reagent to catalyst and even precursor for materials science explorations. It’s that rare blend of stability, versatility, and accessibility that lets chemists lean on it for both fundamental research and specialized industrial use.
Physical & Chemical Properties
Triphenylbismuth carries a molecular formula of C18H15Bi and tips the scale at 440.29 g/mol. You’ll notice its subtle yellow crystals, whose melting point sits comfortably around 72–73°C—a relatively low number for such a bulky compound. Solubility matters in the world of chemical handling; triphenylbismuth dissolves in a variety of organic solvents, especially benzene, chloroform, and ether. Its insolubility in water, on the other hand, keeps it from being a spill hazard in lab settings involving aqueous phases. Structurally, the bismuth center draws on its 6s2 lone pair, holding the phenyl rings in a sort of umbrella formation, a shape that hints at its reactivity in transformations and catalysis. This orientation gives triphenylbismuth a well-documented edge for organic manipulations not often achieved with other heavy metal organics.
Technical Specifications & Labeling
Suppliers attach strict labeling to every bottle, usually citing purity (commonly ≥ 98%), batch number, hazard warnings, and recommended storage: dry, cool, and away from oxidizing agents. You’ll find material safety data sheets attached, flagging possible exposure risks. Small traces of bismuth oxide sometimes show up, flagged by reputable vendors and usually posing minimal effect for most roles outside of high-precision synthesis. Handling guidance tends to reflect the relative safety of bismuth compounds, although gloves and goggles stay non-negotiable. Its solid state makes routine laboratory measurement easy, requiring no specialized containment beyond basic chemical hygiene.
Preparation Method
Lab-scale synthesis generally starts with bismuth chloride (BiCl3) and phenylmagnesium bromide (a Grignard reagent), worked up in an anhydrous ether solution. Careful addition and temperature control keep side-products low. The reaction yields magnesium salts and triphenylbismuth, which crystallizes out after solvent removal. Alternative approaches swap Grignard reagents for phenyllithium or use sodium amalgam reduction. On a few occasions, I’ve seen attempts to scale up for kilogram production, which introduce filtration challenges and highlight the need for patient attention to crystallization and purification. Green chemistry groups look into solvent reduction or recyclable phase-transfer catalysts for more sustainable outcomes, but the essential steps haven’t changed much over the decades.
Chemical Reactions & Modifications
Triphenylbismuth brings a unique mix of reactivity—not as hyper as tin or lead analogs, but robust enough for synthetic applications. It can transfer phenyl groups to halides or participate in coupling reactions. You might see it helping out as a ligand source or acting as a mild reducing agent. This chemical keeps surprising researchers with its capacity to undergo oxidative addition, especially with electrophiles; in many hands, it turns into diarylbismuth chlorides or bromides without much fuss. Its use in cross-coupling reactions highlights the nuanced control a heavy bismuth center brings compared to lighter, more toxic heavy metal organics. Newer methods leverage its arylation properties in site-selective syntheses, yielding molecules that help unpick the trickiest routes in pharmaceutical innovation.
Synonyms & Product Names
Triphenylbismuth most often shows up under that name, but catalogues might list it under its CAS number 603-33-8, or call it phenylbismuth, triphenyl-. Some older references still insist on “triphenylbismuthine.” Dark corners of patent literature reference “TPB” or “triphenylbismut” as shorthand. Regardless of label, the structure remains true: three phenyls gripped by a single bismuth atom. Suppliers streamline ordering with these terms, rarely causing confusion in a well-organized laboratory.
Safety & Operational Standards
Health and safety officers breathe easier handling triphenylbismuth compared to lead or arsenic organometallics. It doesn’t vaporize easily, and direct handling rarely causes acute symptoms. Even so, chemical safety standards demand gloves, eye protection, and good ventilation. Long-term data on chronic exposure remains limited, so treating it with the same respect as other heavy metal reagents prevents hard-to-reverse mistakes. Waste disposal follows local hazardous protocols—most facilities collect it with other metal-bearing organics or return it to chemical vendors for safe destruction. For reagent spill response, inert absorbents followed by secure containerization do the trick, preventing environmental release.
Application Area
Chemists lean on triphenylbismuth for a range of synthetic tricks, especially when mild arylation is needed without the side effects of tin or lead. This compound enters the relay race of organic synthesis, photochemical research, and materials development. The pharmaceutical sector taps into its selective reactivity for complex molecule assembly, enjoying its ability to avoid some of the harsher byproducts common with other organometallics. Electronics researchers chase after new bismuth-based semiconductors, pulling inspiration from its structure for precursor design and dopant engineering. Even in catalysis, triphenylbismuth earns citations for facilitating mild oxidation reactions—far less toxic than the chromium salts or stronger oxidizers usually required. I’ve seen academic groups turn to it for both undergraduate demos and cutting-edge research, pleased with its gentle environmental footprint and reliability in the flask.
Research & Development
The past decade saw a surge in research citing triphenylbismuth, mainly as chemists probe beyond tin-centric catalysis for organic coupling reactions. Green chemistry researchers ask how bismuth organics could help phase out more hazardous synthons. Students learn the ropes with triphenylbismuth because its unpredictability is balanced by manageable risk, a perfect mix for training new hands. Research labs tweak its structure, sometimes adding electron-withdrawing or donating groups to the phenyl rings, unleashing new catalytic or electronic properties. Industrial R&D efforts with this compound favor its role as an intermediate in specialty material synthesis, particularly in crafting polymers or high-value electronics. Collaborations between academic and industrial researchers move towards real-life applications in medicine, sensors, and composites, each time peeling back another layer of triphenylbismuth’s potential.
Toxicity Research
Compared to organotin or organolead compounds, triphenylbismuth shows notably lower toxicity in most studies. Acute exposure in animals leads to few irreversible effects at low doses, but chronic toxicity data remains thin. Researchers test its breakdown products to be safe, watching for phenol derivatives and elemental bismuth that might lurk in waste streams. Some environmental experts raise flags about bismuth accumulation, though current evidence pegs its risk well below many other heavy metal organics. Despite this, no one treats it casually; every researcher or technician trained on it learns how quickly chemical fortunes change with dose, route of exposure, or new regulatory findings. Universities and manufacturing plants each run periodic reviews to ensure triphenylbismuth doesn’t slip through the cracks in chemical safety programs.
Future Prospects
Triphenylbismuth sits poised to expand its use as chemists seek more sustainable, lower-toxicity reagents for tomorrow’s synthesis. With the shift away from more hazardous metals, interest in bismuth-organic chemistry continues to build. I expect to see new roles in medicinal chemistry and next-generation materials, especially as researchers design hybrids that couple the organometallic world with biological targeting. Calls for clean and greener synthesis play right into triphenylbismuth’s hands, since its resistance to hydrolysis and air-oxidation fit modern lab demands. The future may even see regulatory easing for bismuth organics, as more data accumulates to support its relatively clean profile. Still, real progress depends on interdisciplinary collaboration—getting the right minds in chemistry, toxicology, and engineering to shape a responsible and effective future for this sturdy compound.
Catalysis in Polymer Chemistry
Triphenylbismuth finds a solid foothold in the production of polymers, especially in making silicone rubbers. Factories have used it to shape and speed up the crosslinking process, which helps mold silicone parts used in everything from bakery mats to automotive gaskets. Compared to old-school catalysts, triphenylbismuth stands out because it shows lower toxicity and works at lower temperatures, making it better for people who handle the process daily. Bismuth-based catalysts also land on regulatory safelists, especially in Europe, where limits on heavy metals push industries to seek cleaner options.
Role in Organic Synthesis
This compound brings versatility into organic labs. Chemists turn to triphenylbismuth as a reagent for many arylation reactions. Making carbon–carbon or carbon–nitrogen bonds in laboratories often takes a lot of heat or careful handling, but triphenylbismuth eases this work. Its role in the preparation of key pharmaceuticals keeps growing. For example, triphenylbismuth helps develop some anti-cancer agents, working alongside palladium-based catalysts to stitch together complex molecules found in the latest cancer drug research.
Application in Electronics and Semiconductors
Most people don't realize that the bright screens and thin devices many use come from careful material science. Triphenylbismuth gets involved in making fine films used for semiconductors and LED components. It acts as a source of bismuth in chemical vapor deposition, helping lay down precise bismuth layers. Companies that fabricate these components benefit from its relatively low toxicity and its ability to deposit thin films smoothly. Over the past decade, as demand for efficient electronics rises, safer compounds like triphenylbismuth became more attractive over older, hazardous options such as trimethylbismuth.
Advances in Medical Imaging and Pharmaceuticals
Researchers have explored triphenylbismuth for imaging work. The heavy bismuth atom shows promise as a contrast agent in X-ray diagnostics. Unlike lead-based materials, bismuth compounds offer less toxicity and better compatibility with the human body. Pharmaceutical groups keep an eye on such innovations as the push for safer imaging tools keeps rising. Some early-stage medicines also use it as a starting material, capitalizing on its mild toxicity and ability to introduce complex aryl groups into drug candidates.
Cleaner Industry with a Focus on Worker and Environmental Safety
Safety marks every turn in modern manufacturing and lab research. Triphenylbismuth stands out for its low toxicity, with far less risk than older bismuth or tin compounds. Workers involved in production lines or R&D labs appreciate the move toward chemicals that cut down acute and chronic hazards. For example, the European Chemicals Agency (ECHA) doesn’t list triphenylbismuth as a substance of very high concern, and groups like OSHA have not flagged major issues in regular handling, provided labs keep up with good ventilation and gloves as standard protective measures.
Tackling Current Challenges
Some challenges hover around the scale of manufacturing and controlling waste. While bismuth is less toxic than tin or lead, large-scale chemical production still creates waste streams. Industry, academia, and regulators need to work together to ensure bismuth recovery and recycling become a bigger part of the story. Government-backed research, incentives for greener production lines, and robust training for chemistry workers all help lower risks as demand for triphenylbismuth continues to climb.
Chemical Formula and Molecular Weight
Anyone familiar with organometallic compounds soon bumps into triphenylbismuth. Its formula—C18H15Bi—gives away its structure plainly. There’s a bismuth atom at the center, flanked by three phenyl rings. That skeleton delivers a molecular weight clocking in at about 419.39 g/mol. Three benzene rings aren’t light by any stretch, and adding a heavy metal like bismuth puts a hefty stamp on the final mass.
Getting to Grips with Triphenylbismuth
Bismuth itself rarely finds the spotlight in everyday conversation. Most people know it from pink medicine or obscure trivia about heavy metals. But in chemical research, triphenylbismuth shows up where people need organometallic reagents with unique reactivity. You can spot it most often in organic synthesis labs and materials research.
In my grad school days, handling triphenylbismuth meant strict respect for lab protocols. It doesn’t explode or burst into flames at a sneeze, far safer than many of its period-table neighbors. Its crystalline powder refuses to dissolve in water. It prefers organic solvents like chloroform or ether, often necessary for chemical reactions.
Why Formula and Weight Matter
Knowing the formula and exact mass isn’t just homework for chem students. Moles and milligrams run every reaction. I learned early on: weigh wrong, waste a day or worse, get skewed results. A compound this dense demands precision. If you start with an incorrect molecular weight, your yields, purities, and downstream steps suffer. It cuts to the core of reliable, repeatable science—nobody gets far on guesswork. Labs keep up-to-date analytical balances and certified reference data on hand because consistency and accuracy outweigh everything.
Triphenylbismuth in Research and Industry
Triphenylbismuth finds itself at home in synthetic chemistry where it helps couple molecules, break bonds, or even act as a source of bismuth in various reactions. The push for sustainable catalysis stirred interest because bismuth’s less toxic than many other heavy metals. It’s still not benign; any heavy-metal compound needs care—good gloves, fume hood, solid disposal programs—otherwise risks build up fast.
People have experimented with triphenylbismuth in making new materials for electronics and green polymers. Bismuth-based catalysts can drive reactions that lower energy bills or trim hazardous waste. Industry doesn’t just chase the new for bragging rights. Meeting modern regulatory demands on toxicity and eco-footprint means reevaluating tried-and-true reagents. Triphenylbismuth may play a part in that shift.
Tackling Challenges and Embracing Solutions
Use of any heavy-metal organometallic demands rigor. In some labs I worked in, tracking the flow of bismuth-laden waste was almost a sport. The trick lies in combination: smart process engineering, closed system reactors, and robust training all pull together. Partnering with certified hazardous waste handlers ensures compliance and safety for the team and community. There’s never full escape from risk but constant attention brings it down to manageable levels.
Research and development heads looking to the future treat triphenylbismuth as a tool, not a toy. Applications get weighed against risk and regulatory cost. Tight collaboration between chemists, EH&S teams, and supply chain experts has become the norm.
Closing Thoughts
Understanding the structure and mass of triphenylbismuth goes beyond the trivia of chemical notation. Informed decision-making—guided by not just chemical formulas but their real-world implications—builds a stronger, safer research culture and industry. Knowing the numbers serves as the foundation for every safe, effective, and innovative use of compounds like triphenylbismuth.
What Is Triphenylbismuth?
Triphenylbismuth crops up mostly in specialty chemistry labs and a handful of manufacturing settings. It looks pretty unassuming at first glance—a solid chunk, almost crystalline, sometimes with a faint musty odor. You won’t find it in your kitchen, but it sneaks into important places, including electronics and pharmaceutical production. Digging into the safety side, the catch is usually not what it does in the finished product, but what it means for the folks who spend their time with this compound day after day.
What Science Says About Its Toxicity
The word “bismuth” can make people think of the stuff in pink medicines or specialty cosmetics, which usually rates as safe. Triphenylbismuth stands out here—it has three phenyl rings tacked on, and that difference matters. Bismuth metal by itself is pretty tame. Chemists even tell stories about eating bismuth chunks as a party trick. But connecting bismuth to phenyl groups changes the way your body deals with it.
Animal studies point to some low-level toxicity with triphenylbismuth. Inhaling dust, getting it on your skin, or swallowing it can irritate tissues. The tough part? The long-term consequences of repeated exposure haven’t been mapped out. In the 1990s, workers handling aromatic bismuth compounds faced stricter health checks after researchers found some versions built up in organs and fiddled with red blood cell chemistry. A rat study from Japan logged kidney stress in animals dosed with triphenylbismuth—but the same study didn’t show dramatic damage unless you upped the dose far past workplace levels. Still, safety data sheets for triphenylbismuth don’t sugarcoat it: gloves, goggles, and good ventilation matter.
Why Hazard Levels Vary By Use
Sparking a chemical reaction or working up a new batch of electronics materials, chemists might use a few grams of triphenylbismuth. Typical customers wear protective suits, know the safety drills, and treat it with respect. In my small-scale research experience, lab managers kept a tight lid on anything with bismuth in the formula. It’s not radioactive or explosively toxic, but it isn’t casual-cook chemistry either. The real safety problems can creep in when cleaning up spills or dumping waste. Pouring leftovers down the sink is a quick way to invite local water contamination and some awkward questions from inspectors.
People get nervous around any chemical with “phenyl” in the name, and that’s justified—a lot of them can pass through skin or break down unpredictably. Triorganometallics, including triphenylbismuth, deserve more respect than bismuth alone. Regulatory agencies like OSHA and the European Chemicals Agency keep these compounds on their watchlists, even if the documented human poisonings are rare. Personal experience says most chemists would rather handle bismuth powder than its triphenyl cousin, knowing the risk comes from regular contact.
Reducing the Risk: What Can Help?
Rather than tacking on blank warnings, companies can stay ahead by switching to closed handling systems and step up personal training. Spill kits designed for organometallics cut down the mess right away. Medical checkups for workers, especially those in electronics and chemical manufacturing, help catch issues early. Waste needs careful labeling and a trusted disposal partner so nothing backfires down the road. Every year brings more eco-friendly catalyst options—sometimes, making the switch cuts health concerns at the root. Triphenylbismuth might not sit next to cyanide on the danger scale, but brushing off basic precautions can nudge workplaces into trouble fast. Proper respect, decent engineering controls, and regular check-ins give peace of mind when working with compounds outside the safe-and-gentle category.
Why Triphenylbismuth Deserves Attention
Triphenylbismuth shows up in many chemical labs as a catalyst or ingredient. Folks who work with organometallics bump into it more often than most. It doesn’t just sit on a shelf; safe handling and storage matter a lot. Years in the lab taught me that even substances that seem harmless can bite back if you get complacent. Bismuth compounds might not grab headlines for toxicity, but ignoring basic precautions leads straight to trouble.
Real Hazards Lurking in the Details
Triphenylbismuth usually forms as a white to off-white powder. It’s not an explosive, but it reacts with strong oxidizers, bringing fire hazards into the picture. Some people may shrug off any danger, thinking—wrongly—that bismuth ranks among the “non-toxic” elements. Sure, it’s less dangerous than heavy metals like lead, but inhaling dust or touching bare skin opens you up to unknown risks. Manufacturers warn about irritation to the eyes, skin, and throat. NIOSH and other safety bodies echo these concerns, especially for repeated exposures.
Smart Storage Makes All the Difference
One lesson I learned from cleanup duty—store chemicals with an eye to their long-term behavior. Shelves stacked close to heat, direct sunlight, or moisture turn powders like triphenylbismuth into messy, unpredictable substances. Glass bottles with airtight caps never failed me, as long as they stood in a dry, cool cabinet away from acids and oxidants. Any chemical that picks up moisture clumps or cakes, turning scooping into a mess that clouds the air with dust.
Proper labeling helps everyone sleeping better at night. Even if you know every bottle by sight, someone else might step in and not realize what’s there. Nothing makes a supervisor wince faster than mystery jars left behind in an old drawer. Clear hazard labels and updated inventory mean no mix-ups, even during a shift change.
Handling: Gloves, Eyes, and Ventilation Matter
You don’t want to learn about chemical burns by accident. Always reach for gloves—nitrile or neoprene gives good protection. I caught some folks wiping spills with bare hands, then complaining about rashes later. Good goggles matter, even if you think a quick transfer isn’t risky. Lab goggles with side shields hate surprises, and a face mask or dust mask never hurts when dealing with powders.
Working under a well-maintained fume hood wins every time. Once I ignored this step, tried to weigh triphenylbismuth on the bench, and ended up coughing for days. Good airflow pulls dust away before it hits your face.
Accidents do happen. Keep eye wash stations within reach and make sure everyone knows where the safety showers are. Early action beats regret any day.
Moving Toward Better Solutions
Training is worth more than a thousand warning stickers. Even resourceful teams slip up if the safety message fades. I saw labs cut incidents in half just by reviewing chemical hazards every few months. Suppliers can help by shipping smaller packages. This way, people only open what they really need, cutting down on dusty cleanups and old, deteriorating stock.
Triple-check spill kits and disposal bins for chemical waste. Improper disposal can send low-level toxins into groundwater or landfills. Responsible handling after use matters just as much as storage on day one.
Triphenylbismuth won’t spark catastrophe with every mistake, but real-world lab problems grow from small oversights. A culture built on vigilance—gloves, labels, fresh air, and honest teamwork—protects people and delivers better results.
Understanding What’s on Offer
Triphenylbismuth isn’t a chemical you see in the headlines, but I’ve seen it crop up plenty in the lab, especially where precision matters. You often find this compound in organic synthesis, electronics, and sometimes, oddly enough, in pharmaceutical research. Whether someone pulls it off a shelf for a catalyst or as a reagent, purity really makes or breaks how well it works for the task at hand.
Most suppliers carry a few distinct purity grades, and there’s usually no guessing game about which suits what job. The basic grade, often labeled as “technical,” usually sits around 97% purity. I’ve worked with this kind for preliminary experiments, where cost and speed win out over perfection. There’s some leeway for things like minor organic or inorganic leftovers. Fine for larger-scale testing or when reaction tolerances run wide, but you start to hit snags if you expect flawless results.
Stepping Up the Game: High Purity, Extra Caution
Demand kicks up a notch above 99%, sometimes marked as “analytical grade” or “reagent grade.” This one’s seen in sensitive pharmaceutical analysis or certain electronics work. Batches in this range often land between 99% and 99.5%. High purity stops unwanted side reactions—always a relief for a chemist watching for one mystery blip to ruin a day’s work. If I’m running a reaction where every atom counts, or if contamination would ruin a batch, this grade pulls its weight.
There’s an even smaller market for “ultra high purity”—pushing to 99.9% or higher. These sorts are rare outside of specialized research, like advanced materials science or microelectronics, because the effort and cost of reaching that level climbs steeply. Still, researchers or engineers can’t compromise if semiconductor reliability or trace analysis is in play. Trace metals, solvents, or leftover organics need strict control. Last I checked, a few European and Japanese producers strive for even higher numbers, but shipping and verification get complicated fast.
Risks When Purity Slips Through
Using the wrong grade usually puts more than the experiment at risk. In electronics, an impurity can short-circuit a device before it ever ships. In medicine, even a trace element out of place risks patient safety or burns months of work. I’ve had to rerun entire trials for missing a grade label—or worse, relying on outdated documentation. Mishaps like these show why tracking and verifying purity isn’t just red tape.
Improving Quality Assurance
There’s enough confusion in the real world already, so a clean supply chain and transparency matter just as much as the label. Reliable certificates of analysis, third-party verification, and batch tracking tools support everyone from the supply side to scientists at the bench. Spot testing using techniques like gas chromatography or atomic absorption can catch small errors early, saving headaches all around. I recommend working with suppliers known for accuracy and follow-through, plus keeping close records on each order.
Global demand for cleaner materials keeps rising, mainly because more industries depend on chemicals performing exactly as intended. Researchers watch for faster, cheaper purification methods to improve yield and purity together. Digital track-and-trace brings a new level of accountability, cutting down supply chain errors before they cost real time and money.