Sodium Selenite: A Closer Look
Historical Development
People have been drawn to selenium for centuries, so its discovery didn’t come out of nowhere. Chemist Jöns Jacob Berzelius identified selenium in 1817, and industry soon found a use for its unusual properties. Sodium selenite, a convenient and soluble salt, came into the picture as science progressed during the nineteenth century. Laboratories and manufacturers realized they could harness sodium selenite in glassmaking for its decolorizing powers. In later decades, companies added sodium selenite to feed, as scientists recognized animals needed trace levels of selenium to function properly. Throughout the twentieth century, the pharmaceutical world studied sodium selenite as a supplement and a tool in cancer and oxidative stress research, making it one of those underrated chemical workhorses that pops up in places few would expect.
Product Overview
Sodium selenite wears several hats. At its core, you’re looking at a salt composed of sodium, selenium, and oxygen, typically seen as colorless or white crystals. These crystals dissolve in water far more readily than they do in alcohol, which suits a range of industrial requirements. Commercial producers sell sodium selenite for use in dietary supplements, animal nutrition, glass manufacturing, pigments, textile processing, and sometimes even as a reagent in laboratories. The manufacturing process must keep a sharp eye on contamination, as the margin for safety in food or pharmaceutical applications gets narrow fast. Decades of trial, error, and regulation led to standardized purity levels, packaging, and documentation.
Physical & Chemical Properties
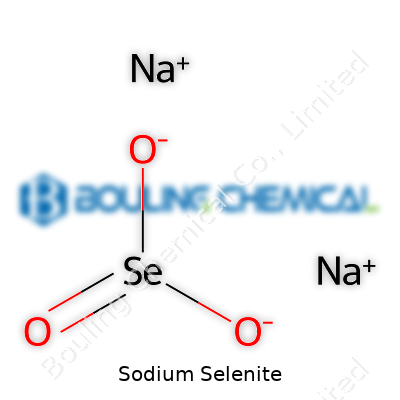
Once sodium selenite sits on your bench, it presents itself as a crystalline solid. Its melting point starts to climb above 700°C, so you don’t see it melting in the lab during normal procedures. It dissolves in water to give a clear solution, but if you spill it on your desk, you get a white, odorless powder that doesn’t attract attention, except for its reputation. In chemical terms, sodium selenite carries the formula Na2SeO3, and it reacts as a source of selenium in the +4 oxidation state. Its oxidation and reduction behavior leads chemists to use it when they want selenium’s biological activity, but without some of the volatility of selenium dioxide gas. In air, sodium selenite stays pretty stable, and moisture won’t break it down quickly, which helps in transportation and storage.
Technical Specifications & Labeling
Industrial and academic buyers demand exacting specifications for sodium selenite, since even slight contamination can undermine entire research or production batches. Pharmaceutical grade material registers minimal heavy metals, with strict caps on lead, arsenic, and related elements. Labels spell out batch numbers, manufacturing dates, expiration dates, and purity, with the most reliable suppliers providing certificates of analysis. Handling and storage instructions appear right on the package due to the compound’s well-known toxicity—protective gloves, goggles, and careful ventilation aren’t suggestions, but requirements, checked by safety officers and inspectors. Whether destined for livestock feed or as a micronutrient in a human supplement, labeling standards help ensure transparency and trackability. Regulatory authorities follow labeling closely because accidental overexposure to selenium compounds gets serious quickly.
Preparation Method
Making sodium selenite isn’t as simple as mixing a few household chemicals. The most common path starts with elemental selenium, usually derived as a byproduct of copper refining, and sodium hydroxide. The process demands precise temperature control and careful addition of oxidizing agents, such as hydrogen peroxide or sodium nitrate, to steer selenium into the right oxidation state. The resulting solution is filtered, concentrated, and crystallized to pull out pure sodium selenite. Quality checks at this stage confirm no carryover from raw materials, especially from copper, sulfur, or impure selenium. Big facilities engineer-out errors with robust controls, yet even small runs in laboratories call for careful purification and diligence, since even minor contamination risks everything from ruined glass color batches to livestock health.
Chemical Reactions & Modifications
Chemists value sodium selenite not just for what it does, but for how it interacts. It serves as a precursor for other selenium compounds, such as sodium selenate, formed by further oxidation. Reducing agents like sulfur dioxide push sodium selenite to elemental selenium, handy in analytical chemistry for selenium determination. Sodium selenite reacts with acidic solutions to precipitate selenium dioxide; with strong acids, it yields selenous acid. In biological settings, sodium selenite incorporates into selenoproteins, essential for antioxidant function and thyroid activity. Metal ions, especially transition metals, form complexes with selenite ions, and this coordination chemistry opens applications across catalysis and materials research. Reactivity ensures sodium selenite remains more than a simple supplement—its chemistry links it to cutting-edge fields, from electronics to imaging.
Synonyms & Product Names
Sodium selenite hides behind a handful of names, depending on the context. Companies and chemists know it as disodium selenite, sodium selenious acid salt, and EINECS 231-576-9. On regulatory and safety sheets, the term “sodium selenite” dominates, but in trade, customer queries reference synonyms and local product codes. In supplements, plain “selenite” sometimes appears, which risks confusion with selenate or selenious acid. The distinction matters, particularly in dosage and effects. International regulations might show minor spelling changes, but anyone working with selenium compounds keeps a close eye on every name and number linked to sodium selenite, sidestepping mix-ups that could lead to product recalls, research setbacks, or worse.
Safety & Operational Standards
Safety with sodium selenite means policy, not preference. Its ability to support health in trace amounts contrasts sharply with hazards at modest overdoses. Direct ingestion or inhalation can cause symptoms from gastrointestinal distress to neurological damage and death, so industrial hygiene matters from the first minute on-site. Modern production lines use ventilated enclosures, sealed transport, and strict inventory controls. Material Safety Data Sheets detail steps for spills, storage, and personal protective gear—gloves, goggles, respirators in some settings. Regulatory watchdogs, including OSHA, EPA, and their equivalents worldwide, enforce regular audits and restrict disposal, since selenium compounds threaten aquatic life and accumulate in ecosystems. Every safety drill and protocol echoes with incidents from the past—lax handling practices have killed livestock and caused workplace poisonings. Knowing the history compels vigilance today.
Application Area
Sodium selenite shows up where life and industry meet. In agriculture, it addresses selenium-deficient soils, keeping livestock healthy and productive. Feed supplement manufacturers fine-tune doses, since just a little too much can spur toxicity. Human nutritional science relies on sodium selenite to fortify foods and design supplements, recognizing that many diets lack enough selenium for immune and thyroid health. Glassmakers benefit from its ability to counteract greenish tints produced by iron impurities, leading to clearer glass for everything from windows to laboratory glassware. Textile firms use sodium selenite for certain dyeing steps, and electronics researchers work with selenium chemistry to develop new materials with unique electrical properties. Laboratory scientists reach for sodium selenite as a reducing agent, a selenium source, and sometimes as a reference material in analytical techniques.
Research & Development
Scientists aren’t finished with sodium selenite—they’re just getting started. Nutritional biochemistry labs investigate selenite’s impact on cellular defense mechanisms, measuring how small doses affect glutathione peroxidase activity and immune markers. Cancer researchers look at sodium selenite’s pro-oxidant properties, using it to target cancer cells’ vulnerabilities, though the line between therapy and harm stays razor-thin. Analytical chemists pursue faster, more sensitive tests for selenium content in blood, urine, and environmental samples, often using sodium selenite as a standard or a spike. Pharmaceutical developers monitor selenite’s potential in formulations for oxidative stress, but have yet to resolve safety challenges. Environmental chemists study soil and water selenium cycling, constantly refining safe use and remediation strategies. In each field, scientists recognize sodium selenite’s utility and danger, sometimes in the same experiment.
Toxicity Research
Toxicology has taught the world that selenium’s window between deficiency and toxicity is tighter than most micronutrients. Sodium selenite enters the body more readily than less soluble selenium sources. In animals, acute overexposure leads to symptoms from hair loss to respiratory failure, and chronic exposure damages organs and upsets reproductive systems. Case studies from livestock operations illustrate how feed mixing errors spark outbreaks of selenosis. Human toxicity, though rare, prompts regulatory bodies to keep dietary selenite intakes below 200 micrograms per day. Modern research explores not just how selenite harms the body, but why some tissues remain more resilient. Newer studies in cell lines and animal models search for early biomarkers of toxicity, aiming to spot trouble before clinical symptoms appear. Dietary guidelines, storage instructions, and handling practices come out of decades of sometimes painful lessons learned in fields, factories, and clinics.
Future Prospects
Looking ahead, sodium selenite will stay relevant as science peels back the role of trace minerals in health. Advances in genomics and proteomics suggest personalized nutrition might one day include precise selenite prescriptions based on genetic makeup and metabolic needs. New delivery forms, smart feed supplements in agriculture, and tighter regulatory oversight could reduce risks of deficiency and toxicity alike. On the industrial side, innovations in glass, pigment, and electronics manufacturing could give sodium selenite new life, especially as demand for specialty materials grows. Researchers hope to unlock its full biological potential, finding uses in aging, neuroprotection, and immune health, but safety and environmental stewardship will drive policy and practice. The story of sodium selenite, shaped by discovery and caution, shows how an unassuming compound can hold both promise and peril for the future.
What Is It?
Sodium selenite stands out as a simple chemical compound, made from selenium combined with sodium. Most folks don’t think about it much, since you won’t spot it on a sandwich or mixed into your morning oats. Still, even though it shows up in tiny amounts, this mineral source shows up in lots of places that might surprise you.
The Role in Human and Animal Nutrition
Doctors and farmers both reach for sodium selenite, but for different reasons. Nutritionists know selenium helps the body stay healthy–it keeps the thyroid running and offers strong antioxidant protection. Here in the U.S., soils in some areas carry nearly enough selenium, but places like parts of China and Europe have seen trouble with deficiency. Instead of letting livestock or people go without, farms and food producers sprinkle sodium selenite into animal feed and fortified foods, making sure diets don’t come up short.
Having grown up around cattle ranches in Texas, I saw firsthand why selenium gets mixed into feed rations. Ranchers watched for signs of “white muscle disease,” a kind of wasting illness triggered when livestock can’t grab enough selenium from grazing. Just a pinch of sodium selenite in the daily ration turned things around, and cattle bounced back. Research backs this up, showing performance and survival rates jump when farmers stay ahead of deficiencies.
Food Fortification in Everyday Life
Sodium selenite doesn’t pop up on ingredient lists as often as salt or sugar, yet it can hide in common foods. Bakers and cereal makers add it to products aimed at people who live where local produce falls short of minerals. Government guidelines don’t leave this to chance—the FDA in the United States, for example, gives the nod to selenium supplementation in meals sold through schools and food programs. European health agencies recognize the same need, especially in regions where crops don’t pick up enough selenium from the dirt.
Medicine and Health Supplements
You’ll spot sodium selenite inside some over-the-counter vitamin or mineral supplements. Doctors sometimes turn to selenium as part of therapy for patients facing thyroid disorders, weakened immune systems, or certain cancers, but the dose matters. Too little, and the body struggles; too much, and selenium crosses into harmful territory. Studies warn about a fine line—nail and hair changes, or nerve problems show up if someone takes in way more than needed. Doctors use blood tests to help figure out what’s missing before recommending a supplement.
Industry Uses: A Small Player with Big Impact
Beyond farms and pharmacies, sodium selenite finds odd jobs on factory floors. It acts as a glass colorant and helps manufacture pigments for plastics and ceramics. Manufacturers use it sparingly since it can irritate skin or lungs. People working with sodium selenite rely on safety training and gear because nobody wants a rash or cough at the end of the week.
Straight Talk on Risks and Solutions
Access to selenium matters most in places where natural levels dip too low. Community health programs track soil and water content and can head off problems with targeted supplements. It’s a balancing act—too little creates problems, while too much threatens public health. Responsible food fortification and farm practices play the main role in keeping populations healthy without tipping the scale. Regular soil tests, food surveys, and transparent labeling help folks know what’s in what they eat and what their animals get.
Fact: Selenium’s recommended intake is about 55 micrograms each day for adults, and sodium selenite makes meeting this goal easier where nature falls short.Understanding Sodium Selenite in Everyday Life
You’ll spot sodium selenite on the back of multivitamin bottles or in some processed foods. Those words often spark quick searches and worries, but this is a real story about a trace mineral playing an important role in our diet. Selenium forms a big part of typical nutrition conversations, mostly because the human body can’t make it on its own. There, sodium selenite steps in as one of the ways scientists make selenium easier to add to food and supplements.
What Science Knows About Safety
Let’s get down to the basics: sodium selenite helps people meet the daily selenium requirement, set at about 55 micrograms for adults. The human immune system, thyroid function, and antioxidant defense lean on selenium to keep things running. Food sources like Brazil nuts, seafood, eggs, and grains already carry this mineral. Still, processed food producers sometimes add sodium selenite to make sure no one misses out.
Problems don’t show up until somebody consumes too much. Short-term exposure to high levels of sodium selenite can trigger stomach pain, nausea, or hair loss. Massive overexposure may bring about something called selenosis, which shows up as skin rashes, dizziness, or nerve problems. The real trouble starts at doses that go way beyond typical supplement or food fortification amounts.
Lean on evidence from public health authorities for a better grip on safe intake. The United States National Institutes of Health set the tolerable upper limit for selenium at 400 micrograms a day for adults. Usual amounts supplied by multivitamins or mineral tablets don’t come close to hitting that ceiling.
Quality and Oversight in Manufacturing
Safety gets a boost from fair labeling and good manufacturing standards. Regulatory agencies such as the U.S. Food and Drug Administration monitor the quality of dietary supplements closely, so the sodium selenite you find in stores usually gets checked for content, purity, and labeling. Companies with a strong record for quality control make a real difference—I’ve seen supplement makers use third-party verification to catch any unwanted contaminants or inaccurate dose claims.
Risks, Vulnerable Groups, and Smart Intake
Most people don’t need to worry about piling up too much selenium unless there’s overuse of supplements or weird eating habits. People living in areas where the soil holds little selenium may actually benefit from fortified foods. Still, there’s no substitute for thoughtful, doctor-led advice. I’ve run into people who tried to “biohack” their diets with mega-doses of every vitamin or mineral, only to wind up with far more problems than they started with.
Pregnant women, small children, and anyone coping with kidney or thyroid conditions should be extra cautious about supplement use. Selenium builds up over time, so a little too much every day can stack up and cause health issues months later.
Better Choices and Smarter Nutrition
Safe nutrition involves choosing well-labeled supplements, following recommended doses, and trusting information from medical and scientific sources. Sticking to official serving sizes matters. The label and the experts matter more than hype, trends, or promises of miracle cures. No nutritional silver bullet exists—instead, a varied diet backed by steady habits works better than any single supplement, sodium selenite included.
The Ups and Downs of Sodium Selenite
People turn to sodium selenite for all sorts of reasons. Doctors sometimes suggest it for folks who aren’t getting enough selenium. It shows up in animal feed and even makes its way into dietary supplements. But every supplement has a flip side. Knowing that sodium selenite comes with its own risk is just smart living. Too little selenium in your system brings trouble, but getting too much won’t do you any favors either.
The Body’s Reaction at Normal and Higher Doses
The body treats sodium selenite as a trace mineral—good in small amounts, not so friendly in large ones. A healthy diet packs plenty of selenium for most people. Soil and crops hold it, and it trickles down to grains, meats, and eggs. Overdosing rarely happens by accident. Issues pop up most often when supplements or industrial chemicals get involved.
Spotting Side Effects: Short and Long-Term
Common problems from taking too much sodium selenite start with nausea, diarrhea, and pain in your gut. You may sweat more and feel cranky or tired. Sometimes your breath carries a garlicky odor, and fingernails develop odd white spots or start breaking. I’ve seen people blame a bad day on stress or sleeplessness, but these symptoms can link right back to too much selenium in the mix.
Long-term or high-dose exposure can hit much harder. Selenium poisoning—or selenosis—can leave nerves frazzled and hair falling out. Skin may erupt in rashes, and joints might ache for no clear reason. If someone keeps taking large amounts, doctors report tooth decay, mood swings, and even kidney or liver damage. Selenium’s been around for ages in agriculture, so farmers sometimes notice hoof problems and dull coats in animals who consume too much selenite.
Digging Deeper: How Sodium Selenite Affects Different People
Not everyone handles supplements the same. Children take a bigger hit from overdoses since their bodies are smaller. Pregnant women worry, too, since the right amount matters for a baby’s brain and organs. If you already handle kidney or liver problems, selenium piles up faster, stacking risk higher. Some medicines mess with selenium’s journey through the body, making it easier to tip from healthy into hazardous.
Food sources make overdosing rare, but supplements can sneak up in unexpected doses, especially mixed with other vitamins in multivitamin blends. Labels don’t always spell out exactly what you get, and batch inconsistencies add to confusion. The “more is better” mentality sold at health stores needs a reality check—it doesn’t take much to move from helpful to harmful, and the margin shrinks for folks already eating a balanced diet.
What Can Actually Help: Practical Steps for Safer Use
Reading up before reaching for any supplement pays off, and talking with a healthcare professional is the next move if selenium deficiency crops up in blood work. Experts set the safe limit of selenium around 400 micrograms a day for most adults in healthy circumstances. A single Brazil nut may contain half that dose or more, so overdosing sneaks up fast if you add supplements to a rich diet. Anyone thinking about sodium selenite pills for “wellness” has a duty to double-check with evidence-based information and measured advice—not just friendly suggestions from the internet.
For people managing long-term health problems or thinking about giving supplements to children, doctor guidance isn’t optional. Monitoring blood levels, asking about food sources, and keeping tabs on unexplained symptoms keeps risk in check. Knowing the line between deficiency and toxicity saves money, time, and a lot of grief in the long run.
What Sodium Selenite Brings to the Table
Sodium selenite, a salt form of selenium, often finds its way into supplements—even animal feeds. Selenium helps the body produce important antioxidant enzymes, like glutathione peroxidase. These enzymes protect cells from damage, one reason this trace mineral keeps showing up in health headlines.
Most folks pull selenium from their plates—brazil nuts, eggs, seafood, whole grains. But foods can’t always fill the gaps, especially in places where soil doesn’t offer much selenium. For some, like people with absorbing trouble due to GI issues or on parenteral nutrition, sodium selenite steps up.
Guideline Dosage for Sodium Selenite
Dietary guidelines help dodge both deficiency and toxicity. According to reliable sources like the National Institutes of Health (NIH), healthy adult men and women need about 55 micrograms (mcg) of selenium per day. Pregnant individuals do best with 60 mcg; lactating folks benefit from 70 mcg daily.
Sodium selenite usually delivers selenium in supplement or injectable forms. Multivitamins for adults in the US typically pack 55-200 mcg selenium. In settings like IV nutrition, doctors often dose sodium selenite at around 20-60 mcg per day, adjusted by age, sex, and health status.
Babies and small children have lower needs—7-20 mcg for infants, 20-40 mcg for toddlers and young kids. Teens need more, up to 55 mcg by age 14. Sticking with healthcare guidance matters since selenium tips into toxicity at high doses. Chronic excess, more than 400 mcg daily, leads to symptoms like brittle nails, hair loss, skin rash, and even nervous system problems.
Why Sticking to Evidence-Based Dosing Matters
Too little selenium leaves the immune system limping. Weakness, difficulty concentrating, muscle soreness—these are just the start. Keshan disease, a heart condition from selenium deficiency, showed up in parts of China in the last century and still rears its head in deprived regions. On the flip side, too much selenium builds up quickly. Trust me, nail changes and stomach upset aren’t worth chasing vague promises of energy or “detox.”
Research on selenium and chronic disease gets a lot of attention. Some early data linked selenium with lower cancer risk, mostly in areas with low soil selenium. Later studies, including those funded by the National Cancer Institute, didn’t repeat that benefit, and sometimes saw increased risk at high intakes. So the science keeps evolving, but the basic message stands: get enough—but not too much.
What Informed Dosing Looks Like in Real Life
In my experience, people like clear numbers. But bodies aren’t machines. Some individuals break down nutrients slowly, others quickly. Genetics, medications, and even how much processed food enters the routine shape selenium needs.
Checking with a doctor or registered dietitian matters. Many patients ask about adding selenium during colds or after reading about thyroid health. Blood tests can reveal deficiency or excess. Doctors can fine-tune supplements, especially if underlying issues affect absorption.
Sodium selenite plays a role when food choices fall short or medical conditions demand. As always, the safest choice starts with food and ends with individual guidance—taking just enough for health, and not a sprinkle more.
Looking Beyond the Label
You’re reading the back of a supplement bottle and sodium selenite pops up. Selenium sounds familiar – it’s known as a trace mineral the body needs. So does it make sense to take sodium selenite for your daily dose?
Selenium: More Than Just a Buzzword
Selenium helps with certain enzymes, the immune system, and even the creation of thyroid hormones. People usually get enough through food like Brazil nuts, seafood, or eggs. Still, some look to supplements for an extra boost, especially if they live in places with low selenium in the soil or have diets with limited variety.
Sodium selenite acts as one form of selenium you’ll see in multivitamins and standalone supplements. The reason manufacturers use it comes down to cost and shelf life—it’s stable and budget-friendly compared to some organic forms. You also see it used in animal feed and fortified foods.
Why It Matters: Getting the Dosage Right
The body deals with different selenium forms in its own way. Studies point out that sodium selenite absorbs well enough. The twist is what happens if you get too much. Selenium is one of those nutrients where dose swings from "enough" to "too much" pretty quickly, and going over the recommended intake can lead to problems. Common symptoms include hair loss, GI problems, and more serious risks like nerve damage or issues with the heart. You’re looking at a recommended daily dose of about 55 micrograms for adults, and things start to look dangerous around 400 micrograms a day.
Not all supplements clearly mark the form of selenium they use, and some people think that because sodium selenite feels “chemical,” it might not be as safe or useful. Science, though, hasn’t drawn strong lines between the forms—at least for basic supplementation—after all, the human body evolved handling everything from food. No one sells selenium-enriched nuts that only use one chemical form.
Risks and Responsibility
In my experience, the real trouble starts when someone thinks more is better, especially with minerals. People often forget these aren’t vitamins where the body gets rid of extra amounts so easily. Selenium build-up is a real thing if you double up on supplements, take it with fortified foods, or eat a high-selenium diet already.
The U.S. Food and Drug Administration keeps an eye on supplements but doesn’t enforce the same checks as prescription drugs. This means quality control isn’t always guaranteed, and mistakes happen—a scary thought when you’re talking about nutrients with a slim margin for safety. I once heard about a supplement batch where the levels ended up ten times higher than what appeared on the label. That left a lot of folks with selenium poisoning.
Better Choices and Smarter Habits
The easiest solution is to get your minerals from food most of the time. A couple of Brazil nuts—or a serving of tuna—does more than a synthetic pill. For folks with gut conditions, poor diets, or on dialysis, a doctor can run checks to see if a supplement makes sense. Anyone using supplements should stick to the label and not layer selenium sources without a doctor’s advice.
An open conversation with your health provider matters more than clever marketing. It’s also good to check if consecutive products you’re using—multivitamins, protein powders, fortified cereals—combine to push selenium above safe levels. Trust your body more than bold labels.