Sodium Selenate: A Closer Look at Its Past, Present, and Future
Historical Development
Sodium selenate entered the world’s chemical catalog in the late nineteenth century, tracking alongside modern interest in trace minerals. Early researchers, driven by curiosity about selenium’s role in health and soil composition, stumbled onto the compound during efforts to separate selenium from tellurium. As industry demanded more precision, sodium selenate found its way into glass manufacturing and agricultural experiments, especially during times when selenium deficiency showed up in livestock. Researchers working on nutrient cycles noticed selenium’s ability to boost plant growth in seleniferous soils, further bumping up sodium selenate’s profile and broadening its applications.
Product Overview
Today, sodium selenate often appears as a white, odorless, highly soluble powder or crystalline solid. Chemists and plant scientists prefer it for its reliable selenium content and easy handling. Packaging often lands in sealed, moisture-proof containers, reflecting its hygroscopic nature. The product frequently enters the market at high purity grades—up to 99 percent—catering to pharmaceutical and agricultural customers with rigorous standards. Formulators appreciate its robust shelf life and stability when stored under appropriate conditions.
Physical & Chemical Properties
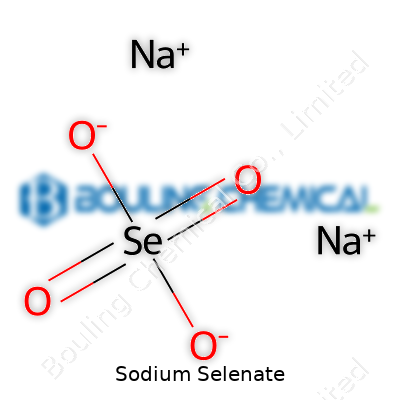
Pure sodium selenate holds the formula Na2SeO4, with a molecular weight of about 188 g/mol. It dissolves in water, giving clear, colorless solutions even at ambient temperatures. The crystalline solid melts at roughly 196°C, and it carries a slightly salty, bitter taste. It doesn’t volatilize under normal conditions and doesn’t really break down at room temperature—though it decomposes if heated strongly, yielding selenium dioxide and sodium oxide. Like its cousin sodium sulfate, sodium selenate acts as a strong oxidizer, which explains the caution labels you see on laboratory bottles.
Technical Specifications & Labeling
Quality standards guide sodium selenate production. Most suppliers publish assay values above 98 percent, keeping heavy metals and insoluble matter below low parts per million. Moisture, chlorides, and sulfates receive even stricter control due to their impact on downstream processes, especially when working in animal feed or pharmaceutical settings. Labels flag the compound’s toxic potential, include hazard and precautionary statements, and indicate storage recommendations to minimize accidental exposure and moisture uptake. Safety Data Sheets lay out handling protocols, first aid measures, and regulatory compliance, covering everything from GHS pictograms to local environmental reporting rules.
Preparation Method
Manufacturers typically start from selenium dioxide—produced by roasting elemental selenium in air—and dissolve it in sodium carbonate or sodium hydroxide. Once the solution cools, sodium selenate crystallizes out upon evaporation or cooling. Large-scale synthesis favors batch reactors, where temperature, pH, and concentration get monitored all along the way. Some setups add filtration and recrystallization steps to drive purity up. Waste management plays a big role; leftover selenium from synthesis can harm aquatic life, so recycling loops sometimes reclaim selenium for use in future batches, reducing environmental impact.
Chemical Reactions & Modifications
Sodium selenate participates in reduction reactions, converting to sodium selenite or even elemental selenium under controlled conditions. In agriculture research, the compound gets reduced in soil by natural microbes, transforming into less soluble, less bioavailable forms. The oxidative strength of sodium selenate also sees play in organic syntheses, especially for preparing organoselenium compounds in medicinal chemistry labs. Unwanted side reactions usually tie back to its strong oxidizing character—something that poses fire risks when mixing with combustible or easily oxidized materials.
Synonyms & Product Names
Trade and scientific communities know sodium selenate under several aliases. On chemical order forms, it might show up as disodium selenate or selenium sodium salt, often with CAS number 13410-01-0 attached. Pharmacopoeias and fertilizer catalogs sometimes note the heptahydrate variant, called sodium selenate-7-water or Seledrex in certain markets. International suppliers stick to these names for standardization, supporting traceability across research, pharmaceutical, and feed industries.
Safety & Operational Standards
Handling sodium selenate safely means embracing strict protocols. Workers in formulation or packaging labs wear gloves, goggles, and dust masks, even if exposures seem fleeting. Ventilation systems keep dust levels low, and strict hygiene stops ingestion through contaminated food or cigarettes. Spills receive immediate cleanup, shifting to wet wiping instead of sweeping to avoid airborne dust. Training goes deep on toxicity signs—garlic breath, gastrointestinal upsets, neural symptoms. Emergency showers and eyewash stations sit nearby, and disposal steps follow hazardous waste procedures to the letter. Storage takes place in cool, dry spaces away from organic or easily oxidized materials, with regular audits ensuring compliance.
Application Area
Plant nutrition claims a big share of sodium selenate’s market. Agronomists use small doses to enrich grains and forage crops, especially in selenium-poor soils in regions like Finland and parts of China. Farmers lean on it to combat “white muscle disease” in sheep and cattle, reflecting its status as an essential micronutrient. Beyond agriculture, sodium selenate acts as a selenium source for dietary supplements, though only under closely regulated doses. Glassmakers sometimes dope specialty glasses with selenium compounds for color control. Researchers in chemistry and materials science pick it up for synthesizing selenium-containing compounds with potential medical or electronic uses. Trace analysis labs dissolve sodium selenate as calibration standards for spectrophotometry, using its known selenium concentration to validate their methods.
Research & Development
Studies over the past decade tapped sodium selenate for roles in health, from managing metabolic disorders to targeting cancer and neurodegeneration. Work at universities in Australia and Europe explores its effect on tau protein in Alzheimer’s disease, capitalizing on its role as a protein phosphatase activator. Other labs focus on slow-release forms that deliver selenium without high peak concentrations, aiming for easier mixing into animal feeds and improved bioavailability. Molecular biologists test selenate’s influence on selenoprotein expression, mapping out the genes and regulatory networks involved. Ongoing projects eye environmental impacts, especially in run-off scenarios from selenium-fortified fields—helping policymakers balance soil health with ecological risks.
Toxicity Research
Dose makes the poison with sodium selenate. Studies dating back to the 1940s flagged how narrow the margin is between dietary necessity and toxicity. In people and livestock, low levels foster cellular health and antioxidant defense, yet slight overshoots trigger nasty symptoms—hair and nail loss, skin lesions, neurological issues. Acute exposures at higher doses kill cells by disrupting mitochondrial function and boosting oxidative stress. Regulatory agencies cap selenium content tightly: 0.1 mg/kg for water, about 0.3 ppm for animal feeds. Lab research follows up with chronic exposure models, tracking accumulation in tissues and uncovering possible links to reproductive harm or developmental delays. Scientists have yet to tie low, measured selenate supplementation to increased cancer in people, which keeps debate going about ideal target levels, especially as dietary patterns shift.
Future Prospects
Where sodium selenate goes from here depends on both new technology and health trends. As populations age and diseases like Alzheimer’s or diabetes draw attention, expect a growing push to pinpoint safe, effective micronutrient delivery systems—possibly leading to selenate-based supplements or functional foods. Meanwhile, pressure to green-up agriculture could drive research on selenium-efficient crops or timed-release selenate formulations. Questions about selenium’s environmental cycling will stick around, given the complex chemistry and tightrope walk between benefit and risk. As analytical tools sharpen and selenate’s molecular influence grows clearer, researchers and regulators will keep debating its best uses, balancing practical gains with a watchful eye on toxicity and sustainability.
A Small Compound With a Big Reach
Sodium selenate rarely shows up in everyday conversation, but it touches many corners of modern life. Workers in agriculture, nutrition, and industry know it as more than just a chemical formula. This compound steps onto the scene as a trace mineral, an animal feed ingredient, a research tool, and even a topic of medical curiosity.
Bringing Selenium to Crops and Livestock
Selenium deficiency in soil causes far-reaching trouble, from weak grains to unhealthy herds. Farms in New Zealand, parts of Europe, and some stretches of North America have seen what happens when pastures lack this trace mineral. Adding sodium selenate to fertilizer mixes can keep animals and people safe from a long list of health problems. Cows and sheep grazing on selenate-rich pastures carry healthier offspring and produce stronger meat and milk.
This mineral doesn’t only travel through soil. Feed manufacturers depend on it to supplement animal diets, especially where local crops can’t deliver enough selenium. People who work with horses or manage herds know weak muscles, poor growth, and lower immunity all link back to this one mineral. Getting that balance right keeps animals healthier and herds more productive. The irony is, just a little too much can do harm—selenium’s safety margin runs thin. Farmers and animal nutritionists know that regular measurement and careful dosing aren’t optional luxuries. They’re essential to avoid poisoning or deficiency alike.
Sodium Selenate in Human Health
Vitamins and minerals don’t demand headlines unless something goes wrong, but selenium makes the news for its role in human wellness. The compound helps craft antioxidant enzymes that fight cellular stress and keep the thyroid ticking. A handful of supplement makers fortify their products with sodium selenate, keeping claims in line with nutritional targets recommended by medical authorities. Most people eating balanced meals get enough selenium without extra help, but this isn’t true everywhere. In areas with depleted soil, supplements become a real lifeline.
Researchers dive into more ambitious possibilities. Clinical studies explore sodium selenate’s potential for treating neurological conditions like Alzheimer’s, aiming to slow or stall disease progression. Results remain mixed, but this compound sits on the front lines of medical science thanks to its unique actions in brain chemistry.
Industry Uses That Might Surprise
Outside the farm and the lab, sodium selenate pops up in surprising places. Glassmakers blend small amounts into specialty glass to shift color or improve performance. Water treatment teams sometimes use it to pull heavy metals from contaminated supplies, helping protect communities from environmental hazards. Safety experts never let their guard down with this compound—mistakes around it have lasting effects, especially in workplaces without strong safety culture.
Balancing Benefit and Risk
Getting the benefits from sodium selenate takes serious oversight. Oversight doesn’t sit well with everyone, but ignoring it brings trouble. In the 1980s, cases of livestock poisoning in Australia proved that guessing doses carries lethal risks. The lesson echoes in modern training for farmers, scientists, and factory workers. Every use—nutrition, research, manufacturing—warrants careful measurement.
The conversation about sodium selenate is about more than just chemistry. It ties together farming and food, wellness and risk, science and stewardship. Real solutions grow from education, strong regulation, and honest conversations between industry, health professionals, and end users.
Why People Care About Sodium Selenate
Sodium selenate pops up now and then in supplement aisles, animal feed, and even some fertilizers. It delivers selenium, an essential trace mineral for the human body. Selenium helps enzymes do their jobs, supports the immune system, and plays a role in thyroid function. Getting enough selenium isn’t always easy, especially for folks living far from the sea or relying on food grown in selenium-poor soil. So it’s not surprising to see sodium selenate as an additive or a supplement.
How Much Is Too Much?
Let’s not sugarcoat it—selenium can be a double-edged sword. The difference between a healthy amount and too much is not huge. Experts at the National Institutes of Health say most adults need about 55 micrograms per day, and they set the upper limit at 400 micrograms daily. If someone goes past that limit for weeks or months, side effects start piling up. We’re not just talking about nausea. Symptoms range from hair loss and brittle nails to garlic breath, brain fog, and nerve damage.
Sodium selenate is more concentrated and easily absorbed than selenium from food. This makes it effective and risky at the same time. A tiny bit helps keep the body on track, but it’s also easy to overshoot. That’s one big reason why government agencies keep a close eye on supplement labels and fortified foods.
Evidence From Studies
Doctors and scientists have explored sodium selenate’s effects in people and animals. Evidence shows the body absorbs it fast, so blood levels can shoot up in a matter of hours. In one clinical study, researchers tried sodium selenate in Alzheimer’s patients. Most people tolerated it at lower doses, but a few had side effects even below the 400-microgram threshold. Other trials in healthy adults suggest smaller doses rarely cause short-term problems, but nobody knows exactly what happens if you take it daily for years. On the other end, cases of selenium poisoning from supplements sometimes pop up in hospital records.
No Substitute for Food
Eating Brazil nuts, fish, eggs, or whole grains gives you selenium along with a handful of other nutrients that help the body process it. With sodium selenate, you lose the complexity of food. I’ve always found I feel and function best relying on diverse meals rather than betting on a single pill or powder. Most people living in developed countries who follow an average diet usually cover their daily selenium requirements without extra help.
How to Stay Safe
Anyone thinking about selenium supplements should talk to a doctor first. Routine blood work can show if you actually need more, since excess builds up quietly over time. For anyone living in places known for low-selenium soil, getting advice based on local health guidance makes sense.
Researchers see potential in sodium selenate for certain rare conditions and they keep exploring it in the lab, but it has no business being used freely by folks chasing a little extra energy or immune support. Until more long-term studies come out, better to trust your plate and stay cautious about pouring anything labeled “high potency” down the hatch.
Understanding Sodium Selenate and Its Purpose
Sodium selenate pops up mostly in agriculture, animal nutrition, and, in some rare instances, human supplementation. This compound acts as a source of selenium, which plays a big role in keeping both humans and animals healthy. Selenium backs up thyroid hormone metabolism, immune defense, and shields cells from oxidative stress. Still, a gap looms between getting enough and getting too much. Even a slight mistake in dosage can slide over into toxicity, so this isn’t a mineral to guess about.
Expert Recommendations and Real-World Practice
Regular food usually takes care of selenium needs. For adults, most health authorities pin the safe and adequate daily intake of selenium at about 55 micrograms. That figure includes all dietary sources, not just sodium selenate supplements. So, someone with a varied diet, full of grains, nuts, eggs, and fish—selenium-rich foods—rarely needs an extra push from supplements. If recommended by a medical professional, selenium should never pass 400 micrograms a day for adults. Doses above that creep into a danger zone. The National Institutes of Health and World Health Organization both echo this limit.
Sodium selenate turns up in livestock feed, especially for sheep and cattle grazing on soil poor in selenium. Experts in animal nutrition know each animal’s weight and species before setting a dose, and these come with clear, published guidelines from veterinary associations. Overshooting the mark can cause “alkali disease” and other health troubles in animals, showing that precision counts here just as much as in human diets.
Risks, Safety, and Lessons from the Field
It's tempting to think that piling on more selenium could offer greater health boosts, but it’s not like topping up your water glass. High intake from sodium selenate often brings on side effects fast—nausea, hair loss, nerve problems, even selenosis, a chronic toxicity condition. I've seen people buy mineral supplements with good intentions, but skipping lab tests or professional advice lands them in a doctor’s office explaining sudden symptoms. Unlike vitamin C or other water-soluble vitamins, selenium builds up and lingers if the dose keeps climbing above safe levels.
Accuracy matters. Many health issues show up in places where diet doesn't offer enough selenium, and it’s easy to spot the benefits of supplement programs in certain parts of China or New Zealand. Still, one size does not fit all. Age, health status, eating habits, and even geography play roles, so health care providers take blood levels and local soil content into account before ever suggesting sodium selenate.
Seeking Solutions for Safe Supplementation
Sodium selenate has a role in rare clinical treatments and agricultural settings, but the average person shouldn't shop for it without expert advice. The best step usually starts with a blood test, followed by a conversation with a healthcare provider who knows a person’s history and local food options. In nutrition, more information and a careful eye replace guesswork every time. If in doubt, food sources always win out for safety and reliability, with supplementation as a backup only with trusted guidance.
The Downside of Getting Storage Wrong
Sodium selenate crops up in industries from agriculture to chemicals. It carries value, but it’s one of those compounds you wouldn’t want spilling on your hands or seeping into the water supply. Exposure brings real risks — everything from irritation to toxicity that affects people and wildlife. I’ve watched mishaps in facilities where sloppy storage left employees worried and company reputations at risk. Taking shortcuts just piles up trouble.
What Proper Storage Actually Looks Like
Imagine walking into a storeroom and seeing chemicals stacked carelessly on top of each other – some in rusty cans, some without labels. That’s a nightmare in the making. Sodium selenate hates moisture, so skip storage near sinks, windows, or pipes. Use airtight containers made of materials that don’t react with powerful salts. Plastic or glass work. Metal lids without liners sometimes corrode or fail, so check compatibility. Get the labels right: bold print, hazard marks, and the date you received it. Missing information only invites mistakes during busy shifts.
Avoiding Mixing and Cross-Contamination
Chemical storage areas quickly become chaotic without separation. A few years back, I saw a facility load shelves with oxidizers, acids, and bases all in a jumble. A leaky valve turned into a shelf fire – not because of a dramatic earthquake, just because nobody mapped out storage zones. Sodium selenate occupies its own section, never next to acids, reducing agents, or food items. That separation keeps unwanted chemical reactions from happening, especially if a lid fails or a box tips over. A chemical storage chart on the wall beats searching Google when in doubt.
Temperature and Ventilation Matter
Stable temperature keeps compounds like sodium selenate from breaking down or reacting. Sunlight streaming onto packages can heat up containers, which makes moisture creep inside. Find a dark, well-ventilated space with reliable climate control. Good airflow prevents fumes from building up, which not only protects workers but also limits pressure inside storage containers. Cramped, hot closets breed problems that proper upkeep avoids. Places I’ve visited with best records run fans and check for dampness every week — small routines add up.
Emergency Prep Isn’t Optional
No matter how careful the staff, mistakes happen. The safest facilities set absorbent spill kits, gloves, and goggles within arm’s reach. Employees run drills so even the least experienced can handle an accident without freezing up. Inspections every few months reveal faded labels, cracks, or misplaced containers before things escalate. Staff don’t stash eating utensils or drinks nearby, since a single transfer can cause a health incident. I’ve seen good safety culture turn storage from a risk to just another part of the job.
Documentation and Oversight Build Trust
Track inventory and note every use. This oversight ensures no one tries to “borrow” materials for side projects or lets containers go uncapped. I’ve watched strong safety records lower insurance rates and keep government inspectors satisfied — small details reduce legal and financial headaches. Transparency builds confidence among workers and communities; nobody likes secrecy where chemicals are concerned.
Sodium selenate doesn’t demand luxury digs, but it wants respect. Dedicated, labeled containers, airtight storage, sensible separation, routine checks, and open communication stop something useful from turning dangerous. You don’t have to reinvent safety — just copy what works, do it every time, and empower everyone to care about the rules.
No Magic Pill: Sodium Selenate Facts
Sodium selenate may sound unfamiliar to many folks. For most, it’s just an ingredient hiding on supplement labels or in lists tied to agricultural use. It’s a form of selenium, a trace mineral the body needs for thyroid function and protecting cells from damage. Like most nutrients, where the line runs between “beneficial” and “too much” can get thin. It’s true for salt, sugar, and especially for minerals like selenium.
Toxicity: The Line Between Helpful and Harmful
At safe intake levels, selenium supports immune function, fertility, and brain health. The U.S. National Institutes of Health sets 400 micrograms as the daily upper limit for selenium from all sources—including sodium selenate. More than that, and risk of side effects rises. Too much sodium selenate in the diet or supplements can push the body toward selenium toxicity, known in the medical world as selenosis.
The first clues of selenosis tend to show up where you might not expect. Many notice a garlic-like odor on the breath. Later, brittle hair, nail changes, and skin rashes can appear. Some folks feel tired, dizzy, or nauseous. High doses for weeks can even throw off nerve function, bringing on tingling, numbness, or odd sensations in fingers and toes. In very rare cases of severe overdosing, it can hit the heart and kidneys.
Risks Hide in the Details
Sodium selenate usually enters the body through supplements or rarely, by mistake through occupational exposure. For those using selenium for certain medical reasons—there are ongoing studies for diseases like Alzheimer’s—doctor supervision really matters. Dosing gets tricky, and the difference between therapeutic and troublesome is not huge. Individual biology also plays a big role. Conditions like kidney disease make it harder for the body to clear out excess selenium. Some medications, including ones used in chemotherapy, can alter selenium processing in the body. Combining these without close healthcare guidance adds layers of risk.
Agricultural and Environmental Exposure
While rare, farm and factory workers can run into sodium selenate through contact with fertilizers or animal feed supplements. Airborne dust or contaminated water also pose risks, mostly for folks living or working near mining operations or heavy industry. The body’s response to short-term, acute exposure often looks different from long-term, low-dose buildup. Symptoms show up fast with a big dose but may sneak in quietly after years of steady contact.
How Much Is Too Much?
Some may turn to selenium-rich supplements hoping to “boost immunity” or sharpen thinking, especially since the pandemic renewed interest in minerals. Too many products on the market downplay the dangers. Multiple reports and poison control call logs show risk spikes from accidental double-dosing or mixing lots of fortified foods, all pushing intake above safe limits.
What Makes the Difference: Supervision and Source
Balancing the benefits of sodium selenate with its risks takes clear medical direction and real understanding of personal health needs. Blood selenium checks make sense if you take supplements or have a condition altering selenium metabolism. Doctors or registered dietitians can screen for drug-nutrient interactions or underlying risks. For most people getting selenium in a basic multivitamin or through normal foods—fish, eggs, Brazil nuts—it’s enough for health. If you’re considering a supplement containing sodium selenate, looking at both your diet and health status first pays off.