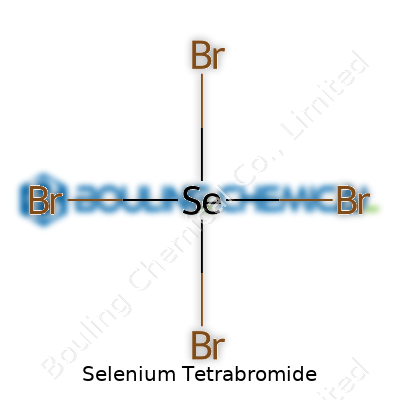
Selenium Tetrabromide: Beyond the Formula
Historical Development
Curiosity has often shaped the way chemistry evolved, and Selenium Tetrabromide offers a good story. Early researchers in the late 19th and early 20th centuries, working with elemental selenium and halogens, started noticing the formation of strange reddish solids during halogenation. Chemists like Berzelius and his contemporaries saw the link between the mysterious crystalline deposits and what we now know as selenium halides. By the mid-20th century, detailed work had nailed down its preparation and physical properties, allowing this compound to move out of the curiosity cabinet and into regular laboratory use. The urge to understand halogen transport, as well as selective oxidation and reduction processes, drove efforts that refined its synthesis. Academic literature in the 1960s and 1970s gave us more systematic data. Today, Selenium Tetrabromide sits as a well-characterized but rarely discussed player in selenium chemistry, overshadowed by its cousin, selenium oxychloride.
Overview
Selenium Tetrabromide is a compound with a somewhat menacing look: dark reddish-brown crystals that sometimes seem almost black. Its formula, SeBr4, tells a story of a selenium atom at the center, surrounded by four bromine atoms. Products like SeBr4 don't win beauty contests, but their chemical curiosity lures both students and experienced chemists into bringing them off the shelf. In labs, this compound rarely serves as a display piece due to its tendency to absorb atmospheric moisture and break down, making safe handling mandatory from the get-go. The tangible feel of working with this compound—solid, dense, with a choking odor—drives home the seriousness of chemicals that combine reactive elements.
Physical & Chemical Properties
Strike a match to the topic of selenium compounds, and most people worry about toxicity and volatility. SeBr4 ranks high on both counts. The solid form melts around 106°C, signaling low thermal stability compared to other heavy element bromides. Water proves a challenge, rapidly hydrolyzing SeBr4 to give off hydrogen bromide and a slurry of selenium oxides. In its purest form, it keeps to itself in dry, airtight containers, crumbling with exposure to moisture in the air. As a Lewis acid, it grabs electron pairs from donors and often features in synthesis pathways with other halides. The compound dissolves in non-polar solvents like carbon tetrachloride, giving reddish solutions that must be handled in tightly sealed gear. Storage demands cool, dark spaces and secure closures to keep the product from breaking down and releasing aggressive fumes.
Technical Specifications & Labeling
Manufacturers label bottles of SeBr4 with a clear hazard statement, warning users about skin, eye, and respiratory risk. The CAS number is 7789-66-4, and one glance at the label brings up warnings about its instability when exposed to moisture. Technical specification sheets offer purity ranges typically above 98%. Chemical suppliers use UN hazard codes, emphasizing requirements for transport and storage. Regulatory compliance shifts by region, but strict labeling—skull-and-crossbones, corrosive markings, and emergency response advice—reflects the global agreement that SeBr4 stands among the more challenging selenium compounds to work with. Even in warehouse settings, chain-of-custody logs prove useful given strict controls.
Preparation Method
The main method of making Selenium Tetrabromide revolves around the direct reaction between elemental selenium powder and bromine liquid. Heating the mixture in a sealed glass reactor, often under an inert nitrogen blanket, kicks off fizzing and bubbling as the dark crystals appear. Some chemists opt for a two-step bromination, starting with selenium dibromide and feeding in more bromine to get the tetrabromide. Large-scale work calls for fume hoods and double-layered gloves, since bromine vapor not only stains but burns. After reaction, the product cools, then gets separated using low-temperature filtration. Slow, careful handling during every step prevents contamination by water or oxygen, which destroy yield and purity. Each batch must get checked for residual elemental selenium and incomplete bromination with titrimetric or spectroscopic analysis, sharpening the prep’s precision.
Chemical Reactions & Modifications
Writers and chemists alike marvel at the way SeBr4 behaves when thrown into a laboratory reaction. It vigorously attacks water and organic bases, forming selenous acid and copious hydrogen bromide gas. React it with strong reducing agents—think iron filings or sodium thiosulfate—and you can coax selenium metal out, leaving behind bromide ions in solution. Reacting it with excess bromine leads to higher selenium bromides, though only in specialized systems. In practice, SeBr4 flashes its utility as a brominating agent, passing on bromines to organic substrates and enabling tricky functional group transformations. Heating SeBr4 splits it into selenium dibromide and molecular bromine, a route some engineers use to manage stoichiometry in multi-stage halide synthesis. Most users tackle these modifications only in sealed, well-ventilated setups.
Synonyms & Product Names
Selenium Tetrabromide crops up under several guises in catalogs and supply chains. Some call it Selentetrabromide or Tetrabromoselenium. In research literature, the abbreviation SeBr4 dominates. A few German and French chemical registries list it as Sélénium Tétrabromure or Selen-tetrabromid, reflecting the compound’s reach across languages and disciplines. Some historic texts refer to ‘Selenium(iv) bromide’ or even ‘Selenium(iv) tetrabromide’, using Roman numerals to point to its oxidation state, but today, the simpler names stick.
Safety & Operational Standards
Anyone who has worked with selenium compounds can recall the caution drilled into every safety briefing. SeBr4 stings eyes, attacks lungs, and leaves burns on bare skin. Fume hoods and face shields stay down for the whole job. Spill kits include not just absorbents but neutralizers for both bromine and selenium waste. Ventilation systems run high as even tiny leaks fill rooms with a harsh onion-like odor. Disposal protocols require double-checking, since waste selenium products often need specialized hazardous waste handling contractors. OSHA and EU standards lay out requirements for protective gear, storage temperatures, and emergency showers in rooms where selenium halides lurk. Training on chemical hygiene—sealing bottles, immediate clean-up, proper documentation—marks the sign of a lab that takes SeBr4 seriously, not just as an experiment but as something that could pose a real health risk with careless use.
Application Area
Selenium Tetrabromide tends to keep a low profile in finished consumer products, but within the lab, it reveals its usefulness as a brominating reagent, especially in situations where both selenium and bromine must enter a reaction in controlled amounts. Some researchers use it as a reference material in the development of selenium analytical techniques, measuring absorption or emission spectra to calibrate instruments. In specialty glassmaking, SeBr4 serves as a selenium transfer agent, lending itself to doping or coloring processes on an experimental scale. Advanced material science teams experiment with it in the modification of chalcogenide materials, tuning semiconducting properties or exploring new synthetic pathways that merge selenium and halogen chemistry. Its use remains tightly bound to labs with highly skilled operators, as few industries find routine need for such a reactive and toxic compound outside R&D settings.
Research & Development
Development work around Selenium Tetrabromide has picked up as chemists look for new ways to manipulate chalcogens for semiconductors and photovoltaic applications. Structural chemists spend considerable effort examining the crystal lattice of SeBr4, chasing clues to unlock better control over halide transport for materials manufacturing. In pharmaceuticals, selenium’s redox properties make it valuable in the exploration of enzyme inhibitors or diagnostic agents, though SeBr4 itself rarely reaches pilot-scale synthesis. Some chemical engineers are testing new, cleaner preparation routes, exploring electrolytic bromination of selenium wastes to recover usable product and minimize hazardous byproducts. Computational chemists run simulations of reactivity, hoping to map out lower-risk synthetic routes before in-lab trials begin. Continued R&D investment aims to squeeze more value from selenium sources, with SeBr4 remaining a critical stepping stone compound in these workflows.
Toxicity Research
Selenium toxicity inspires both dread and scientific rigor. People exposed to SeBr4 can develop selenosis, showing symptoms ranging from dermatitis and gastrointestinal distress to more severe nervous system effects. Animal studies demonstrate rapid onset of tissue damage, particularly in the lungs and liver, reminding workers to steer clear of chronic exposure. Inhalation of decomposition products, such as hydrogen bromide and selenium oxides, deepens risk, prompting agencies to set strict workplace exposure limits. Toxicology studies continue, tracking metabolic pathways and exploring antidotes that blunt cellular selenium overload. Some research teams are probing the difference between organic and inorganic selenium compounds, given emerging evidence that methylated forms follow different toxicological paths. Understanding acute and chronic selenium exposure from SeBr4 helps regulatory bodies set safe design standards and shape workplace policies in laboratories around the world.
Future Prospects
Looking ahead, Selenium Tetrabromide will likely find new roles as researchers push the envelope on semiconductor and specialty glass uses. Progress in green chemistry could drive demand for cleaner preparation and recycling techniques, especially with rising focus on electronic waste. Growing interest in selenide-containing materials for solar cells positions SeBr4 as a potential reagent for doping or thin-film preparation. Toxicity remains a real hurdle, so better containment, detection, and neutralization technologies stand out as priorities. If economic and environmental pressures drive up selenium recycling, expect innovation in the recovery and transformation of selenium halides, including SeBr4. Collaboration between industry, academia, and regulatory agencies will shape best practices and open doors for safer, more efficient use of this fascinating but demanding chemical.
What Stands Out About Selenium Tetrabromide?
Selenium tetrabromide doesn’t pop up in TV commercials or household products, but in a laboratory, it pulls its own weight. The formula SeBr4 spells out a strict, almost fragile balance between selenium and bromine. Anyone who’s ever mixed chemicals in a flask knows that get-it-right-or-start-over feeling. That’s where selenium tetrabromide comes in—most notably in synthesis work. Researchers treat this stuff like a backstage pass to the world of inorganic chemistry.
The Practical Side of SeBr4
Lab professionals value selenium tetrabromide because it acts as a heavy-duty brominating agent. It can step in and swap out atoms in organic compounds, something that opens up new doors when making innovative materials or teasing out new reactions for drug development. For example, when you’re aiming for a specific organoselenium compound, skipping SeBr4 can mean missing your mark. Its powerful bromine content gives chemists a quicker, cleaner way to install selenium atoms in the right spot on a molecule.
In graduate school, stories floated around the lab about how one wrong move with volatile chemicals like SeBr4 could undo hours—sometimes days—of work. That careful approach isn’t for show; exposure to selenium tetrabromide’s vapors can cause headaches and other health issues. Gloves and fume hoods become a second skin. Underestimation has consequences, which is true for any strong brominating agent, but especially for this one.
Why It Matters in the Bigger Picture
Selenium chemistry as a whole often deals with toxicity, sharp smells, and caution tape. The truth is, not many industries outside research and specialty manufacturing reach for SeBr4 on a daily basis. Companies that produce advanced electronics, though, sometimes rely on selenium-based compounds to create semiconductors or special glass with unique optical qualities.
From what I’ve seen, researchers use selenium tetrabromide as a bridge. It links basic starting materials to advanced molecules and products that sit in critical devices—think high-powered solar panels or sensors. Without that link, modern electronics lose an important building block. The trend in materials science keeps chasing better conductivity, more stable finishes, and unique chemical behaviors. Tinkering with selenium and bromine opens up fresh ideas, most of them only possible through reactions that need stubborn reagents like SeBr4.
Trouble Spots and Ways Forward
Handling SeBr4 doesn’t win safety awards. Getting better training, regular hazard drills, and modernizing lab infrastructure help reduce accidents. For example, automatic fume-hood timers and improved ventilation systems play huge roles in keeping chemists safe. Direct experience with spills teaches more than most textbooks do. Personal protective equipment and constant vigilance remain essential.
On the green chemistry front, creators and users of SeBr4 face pressure to find less hazardous alternatives. Many institutions push for innovations that use milder reagents or even predictable, recyclable ones. The transition from research staple to safer substitute isn’t smooth, but every year produces new candidates and methods. So far, none have matched the same combo of power and flexibility as SeBr4, but the search keeps going.
Looking Ahead with Open Eyes
Selenium tetrabromide works behind the scenes in the chemistry world, rarely making headlines outside specialist circles. The value it brings comes with risk, but also with the pay-off of scientific progress. Harnessing its strengths, while staying grounded in health and safety, shows what’s possible when old-fashioned lab grit meets the curiosity to keep chemistry moving forward.
Selenium Tetrabromide: Formula and Core Properties
Selenium tetrabromide turns up from time to time in textbooks and research articles, often written as SeBr4. This specific arrangement means each selenium atom joins up with four bromine atoms. Chemists write it as SeBr4, a pretty direct way to show what's going on at the atomic level. Thinking about it, this sticks selenium—an element closely related to sulfur—in the middle of the halogen action, interacting with four hefty bromine atoms.
Why This Formula Matters in Chemistry
Selenium and bromine combine following clear rules. In my experience working with chemical compounds, formulas like SeBr4 serve more than just the role of a label. They shape lab work, influence experiment outcomes, and guide safety protocols. Exact formulas prevent mishaps. You miss one atom, and your reaction spins off in some wild direction. In a classroom, students may not understand the weight of getting formulas right until they see a stink bomb gone wrong—I've seen bromine mishandled, and nobody in a school lab forgets that headache.
For those designing industrial processes or running tests in research settings, using the wrong ratio of chemicals costs money, burns time, and brings extra risks. The example of SeBr4 reminds us that formulas are more than symbols—they mark the start of everything done with substances in the real world.
Chemical Behavior and Practical Insights
Selenium tetrabromide carries its own quirks. It appears as an orange-yellow solid under normal conditions, reacting with water to release hydrogen bromide and selenium dioxide. That strong, acrid gas from hydrogen bromide isn't something you want drifting across a bench or into a fume hood filter. The formula SeBr4 quickly helps you track the elements involved, spot risks, and plan storage or disposal.
I recall working with similar selenium halides where one misplaced decimal point on a chemical stock sheet upended hours of preparation. The right formula, written in big bold letters on the reagent bottle, catches the eye and saves mistakes. In quality control labs or chemical supply chains, a clear understanding of SeBr4 maintains safety and helps with traceability. It cuts down on confusion when hazards are flagged and cross-checked against international safety data sheets.
Lessons for Science Educators and Industry Professionals
There’s no shortcut in learning chemical formulas—especially for compounds like selenium tetrabromide. For students new to chemistry, formulas like SeBr4 offer a key entry point into understanding structure and reactivity. Teachers who spend time connecting formulas to how things behave in practice help students see past rote memorization. In my own classes, tying formulas to color changes, smells, or safety gear sparks genuine interest in why the arrangement of atoms matters.
On the factory floor or in the warehouse, keeping formulas accurate keeps people safe and operations running. Many firms provide laminated guides, regular safety drills, and software checks to verify just how much SeBr4 they have on hand at any moment. A minor slip in formulas can trigger steel-gray smoke or costly downtime, neither of which any boss wants to explain at a late-night call.
Taking the Formula Off the Page
Selenium tetrabromide, SeBr4, stands as a reminder that the science learned in textbooks finds real-world application every day. From the classroom to the laboratory bench or the factory gate, the chemical formula acts as both checklist and caution sign. Getting it right lays the groundwork for chemistry that works, protects people, and avoids surprises. That level of attention pays off over an entire career in science.
Why Selenium Tetrabromide Draws Concern
Selenium tetrabromide rarely makes headlines, but anyone who deals with chemicals in labs or industrial settings probably recognizes the name. Its striking red-orange crystals signal caution right from the start. Hazard assessments don’t suggest casual handling, and researchers working with this compound always double-check their protective gear before unscrewing the bottle. So, what gives this chemical such a reputation?
Toxicity Isn’t Just a Technicality
It’s easy to miss the risks that step out of specialized textbooks and into real workplaces. Selenium tetrabromide gives off fumes when exposed to moisture in air, and those vapors aren’t something you want to breathe. Inhalation can lead to symptoms like coughing, irritation in the nose and throat, and, at high enough exposures, even trouble breathing. People handling this compound at universities or manufacturing labs sometimes report headaches after even short exposure, and years ago, a colleague had to leave the lab after working with selenium bromides without proper ventilation. The buildup of symptoms gave everyone a wake-up call.
Skin contact doesn’t end much better. Even in small amounts, the substance can burn or irritate. Prolonged exposure ramps up the risks—eventually, you might face skin blistering or severe itching. Lab manuals warn about chemical burns and insist on double-gloving. Splash goggles and lab coats become non-negotiable, not just recommended.
The Toxicity Factor
Toxicity with selenium compounds isn’t limited to direct contact. These chemicals can build up in the body, often targeting the liver and nervous system. The United States Occupational Safety and Health Administration (OSHA) has plenty to say about the strict limits on workplace exposure to selenium compounds. Even low doses, repeated over time, can cause selenium poisoning—otherwise known as selenosis—with symptoms ranging from stomach pain to damage to the fingertips and even nerve disruption. And that’s at exposure levels lower than those that might come from a spill or lab mishap.
Bromine presents its own set of concerns. Adding a bromine element brings risks for corrosive injuries. It can be aggressive with skin and eyes, eating through protective layers if left unchecked. I’ve seen firsthand what happens when bromine chemicals touch unprotected skin—redness appears within minutes, and blisters can form before a person makes it to the safety shower.
Environmental Hazards Matter, Too
Chemicals like selenium tetrabromide don’t stay put. Spills in the lab, improper disposal, or leaks at a workplace can introduce them into water systems. Selenium in the environment can harm fish and birds, and in some cases, poison an entire aquatic ecosystem. Remediation programs after chemical spills focus on selenium’s potential to travel far beyond the initial source. Studies have shown that even a small amount in groundwater can build up over time and disrupt entire food chains. In the 1980s, a US wildlife refuge saw widespread deformities in birds linked to selenium runoff.
Putting Safety and Responsibility First
Safe work with any hazardous material starts with respect and awareness. Training for selenium tetrabromide use centers on airtight containers, fume hoods, full personal protective equipment, and strict protocols for cleanup. Emergency plans—including eyewash stations and spill kits—don’t collect dust; they get regular walkthroughs and practice drills. It’s not about paranoia but about realizing that a few extra minutes of care can avoid weeks of recovery or far-reaching environmental damage. Innovation in chemical safety equipment helps, but everyone’s diligence in daily practice makes the biggest difference.
Looking forward, chemical users and industry leaders would do well to push for even tighter safety standards and more thorough training on subtler compounds. While selenium tetrabromide helps advance science, its risks deserve attention every step of the way. The world doesn’t need another preventable accident or overlooked environmental impact.
Getting Real About Risks
Selenium tetrabromide might not win popularity contests in most labs, but anyone who’s spent time around specialty chemicals remembers the first time they read its label. My hands got a little sweatier than usual. Why? This stuff walks into the room with some heavy baggage: corrosive reputation, strong fumes, and a knack for creating a mess if left unchecked. The hard truth is that plenty of incidents could be avoided just by paying closer attention to storage practices, and not treating every reagent the same.
Why the Dry Route Matters
Selenium tetrabromide reacts fast with moisture in the air. You don’t want to find out what happens if it gets near an open beaker of water or you leave the jar loose on a humid day. I’ve seen some folks cut corners by screwing caps on carelessly. Each time, small leaks caused crystalline gunk to form on the outside of the bottle or, worse, corroded a metal shelf bracket. Besides the mess, you have real exposure risks. So, keeping containers bone dry matters more than just neatness—it’s about your lungs and your skin.
Avoiding the Wrong Company
One lesson that sticks with you in chemical work: not every substance plays nice with others. Selenium tetrabromide loves to raise a fuss if it gets near strong bases, organic acids, or water. I once heard about a minor lab fire that started just because someone stashed it beside sodium hydroxide—careless placement made for a quick scare. It makes sense to use a clearly labeled, stand-alone spot for this compound. Safety data sheets back up what veteran chemists practice: isolation isn’t just paranoia, it’s common sense.
Choosing the Right Shelter
Glass bottles with tight lids, stashed in a chemical-resistant, ventilated cabinet, usually work best. Plastic doesn’t always stand up well to strong bromine compounds—seen too many bottles warp or weaken over a couple years. Metal cabinets seem like a smart idea until corrosion sneaks in. Most labs rely on powder-coated steel with solid liners. The key: keep that storage cool, away from direct heat and sunlight, since heat drives decomposition and starts up those fumes.
Labeling and Vigilance
The words on a bottle can mean the difference between a normal Monday and a trip to the ER. Big, bold hazard symbols help. The bottle won’t walk if someone forgets, but memory isn’t enough—everyone in the room deserves a reminder. Measuring out small amounts in a fume hood, making sure gloves and goggles are not just decorations, checking seals after every use—those tiny habits work in your favor.
Thinking Ahead and Protecting People
Even with the safest shelf, accidents happen. So, spill kits charged with lime or soda ash should stay close, since quick cleanup neutralizes the mess fast. Eye wash stations and emergency showers can save the day if things get out of hand. Team training is the final piece of the puzzle—I saw firsthand how regular drills turned a scary spill into an organized, calm cleanup. That confidence only comes from practice.
Staying on the Good Side of the Rules
Regulatory bodies don’t forgive carelessness with hazardous substances, and they’re right. Local codes call for secondary containment, regular checks, and records of use for a reason. Spot checks keep everyone honest. Chemical safety draws a straight line between good science and good sense. Storing selenium tetrabromide with care turns a risky material into something useful, not dangerous—one good habit and one labeled bottle at a time.
Why Selenium Tetrabromide Demands Respect in the Lab
Selenium tetrabromide (SeBr4) doesn’t show up without a good reason. It’s volatile, it fumes in moist air, and it has a real talent for releasing toxic vapors if you overlook a rule or let your guard down. When I first crossed paths with it during a university project, the pungent odor alone warned me to act with care. A single drop on an unprotected surface sent up yellowish fumes that lingered longer than anyone wanted.
Stories from chemistry departments echo that respect. One colleague learned quickly that a loose glove cuff allowed bromine vapor to reach bare skin, leading to a painful rash. Simple mistakes multiply the risks. These personal tales stick with you far more than any material safety data sheet ever could.
Proper Gear Builds a Wall Between You and Hazards
Think of exposure prevention as building a series of barriers. My standard kit always starts with nitrile gloves layered over cotton, a splash-proof lab coat, chemical goggles, and a full-face shield when decanting or transferring. Some labs go further by using a positive-pressure breathing mask. The brown fumes can penetrate surgical masks in seconds, so investing in proper respiratory protection pays off.
Lab benches need a clear, designated zone for handling. I use polypropylene trays to catch spills because regular glass can etch or crack if the compound reacts with stray moisture or acids. Fume hoods stay on at all times. I never trust simple ventilation: those vapor clouds spread out fast and settle into every nook if left unchecked.
Don't Let Water or Air Mix with SeBr4
Selenium tetrabromide reacts with water faster than you can mop up a spill. The hydrolysis kicks out hydrogen bromide and selenous acid, both highly toxic and corrosive. Even humid air starts the ball rolling. I work only with tightly sealed containers and transfer anything out of its original bottle only inside a glove box under a dry, inert atmosphere, like nitrogen.
Storage always happens in glass with PTFE-lined caps—no rubber stoppers, no metal lids, nothing that could corrode. Fridge storage helps slow decomposition, but keeping it cold isn’t a substitute for keeping it dry and sealed.
Emergency Responses Need Preparation in Advance
Plans for accidental exposures save real pain and injury. Eye wash stations and safety showers must lie within a few steps of the workbench, never blocked by boxes or equipment. Baking soda solutions neutralize acid splashes, but speed wins over all: if someone gets a splash, the solution’s out, and the rinsing starts immediately. For major spills, sand or vermiculite beats paper towels, and cleanup happens using full protection, not wishful thinking.
Disposal isn't as simple as throwing a bottle in the garbage. I package all selenium-contaminated waste in sealed, labeled containers and arrange prompt handoff to professional hazardous waste handlers. Real-world labs don’t take shortcuts, no matter how busy the day looks.
Supporting Every Step with Experience and Science
It helps to remind yourself that missteps cause permanent harm. Agencies such as the Centers for Disease Control and Prevention (CDC) highlight chronic exposure to selenium compounds increases risks of respiratory issues, nerve problems, and skin burns. The science backs up the caution: even low-level exposures add up over time, with effects often showing up months after the fact.
For those working or training with selenium tetrabromide, safety doesn’t come from a checklist. It grows out of habits, consistent preparation, and respect for chemicals that do not forgive mistakes. Peer support and good leadership finish the job, helping keep everyone healthy through every project—no matter how rushed, no matter how routine things start to feel.