The Role of Molybdenum Hydroxide in Modern Science and Industry
Historical Development
Many researchers in the late nineteenth century found their interest sparked by the unique properties of transition metal compounds, notably those containing molybdenum. Early chemists, curious about the mysterious blue-green hues and remarkable reactivity of certain ores, documented the formation of Molybdenum Hydroxide during exploratory syntheses of molybdenum-based salts. These efforts picked up pace in the twentieth century, as scientists began to recognize the practical value of molybdenum compounds in catalysis and corrosion-resistant alloys. After World War II, the demand for better lubricants in mechanical engineering further accelerated studies around various molybdate derivatives, including Molybdenum Hydroxide, leading to a deeper understanding of its chemical flexibility and new manufacturing methods. Over the last few decades, as electronics and materials science advanced, research efforts widened from metallurgy to applications in high-performance catalysts, pigments, and environmental science.
Product Overview
Molybdenum Hydroxide appears as a fine, pale-colored powder or sometimes as a greenish or blue-green solid depending on its precise oxidation state and method of preparation. Its main physical draw—apart from the intriguing color—remains its stability in moderate conditions and amenability to further chemical conversion. Manufacturers rely on its relatively easy preparation and the potential to tailor it for specific applications, whether in chemical synthesis, surface coatings, or specialized catalysts. Chemical distributors typically store Molybdenum Hydroxide in tightly sealed containers, protecting it from excessive moisture and airborne contaminants, since exposure can lead to gradual changes in both structure and effectiveness.
Physical & Chemical Properties
Solid Molybdenum Hydroxide delivers a crucial combination of moderate solubility in acids and insolubility in most organic solvents. Its molar mass varies slightly between its monohydrate and dihydrate forms, roughly centered around 161.95 g/mol for MoO2(OH)2. Crystalline structure often shows up as orthorhombic or monoclinic, which influences performance in catalytic applications. Its density sits modestly above 3.0 g/cm3, which keeps it manageable in storage and transit. The compound boasts good resistance to decomposition at room temperature, yet heats up into complex oxides or even metallic molybdenum under prolonged high-temperature conditions. Molybdenum Hydroxide starts displaying remarkable redox chemistry in the company of reducing or oxidizing agents, giving chemists plenty to explore.
Technical Specifications & Labeling
Producers label pure Molybdenum Hydroxide with tags like “Analytical Grade—Mo content ≥ 60%,” together with clear batch identifiers, country of origin, and detailed handling guidance. Regulatory compliance also matters: products shipped within the United States or Europe require adherence to standards such as REACH or TSCA, with transparent documentation covering any impurities, recommended shelf life, and environmental hazard statements. Labels should describe storage temperatures, outline maximum humidity, and spell out specific precautions for dust inhalation. In laboratory and industrial catalogs, product listings often reference both CAS numbers and UN shipping codes, making sure traceability stays intact from the manufacturer to the user.
Preparation Method
Industrial and laboratory preparation of Molybdenum Hydroxide usually starts from soluble molybdate salts, like sodium molybdate. Acidification with dilute hydrochloric or nitric acid prompts precipitation of the hydroxide. Once the pH reaches the right window, a flocculent solid forms and collects at the bottom of a vessel or on a filter paper. In industry, the process scales up using continuous reactors where solution concentrations, agitation speed, and temperature all stay finely controlled. Operators wash the crude solid thoroughly with deionized water to drive out unreacted ions and byproducts, then either dry the product at low heat or suspend it straight into a secondary process. Some researchers boost yield or change crystalline form by adjusting not just acid type, but also the rate of addition or the presence of chelating agents.
Chemical Reactions & Modifications
Molybdenum Hydroxide exhibits versatility in chemical reactions. It acts as a precursor for higher oxides of molybdenum through careful roasting or calcination in oxygen-rich atmospheres. Under reducing settings, it transforms stepwise to lower oxides, or occasionally even to metallic molybdenum itself. Chemists seeking to modify the material might dope it with other transition metals, or introduce functional groups on its surface by exposure to organosilanes and phosphates. These tweaks shift its catalytic activity, hydrophilicity, or thermal stability, offering fresh directions for energy storage or environmental remediation. Some teams use it to anchor nanoparticles, building hybrid materials fit for hydrogen evolution reactions or pollution abatement. In the academic world, chemical journals brim with reports of Mo(OH)4 derivatives shaped to supply better performance in electrochemical cells or to tackle trace contaminants in water.
Synonyms & Product Names
Outside the lab, Molybdenum Hydroxide goes by a range of synonyms and commercial labels, which sometimes complicates procurement or research. Common alternatives include “Molybdic Hydroxide,” “Molybdenum(IV) Hydroxide,” and “Hydrated Molybdenum Oxide.” Use in catalyst manufacturing or pigment sales might bring up trade names marketed by global suppliers. Catalogs focus on CAS registry numbers to keep confusion at bay, especially since closely related compounds—like ammonium molybdates or molybdic acid—share similar chemistry and physical appearances.
Safety & Operational Standards
Work with Molybdenum Hydroxide brings both rewards and risks. Dust formation generates fine particles, which can irritate respiratory tracts or skin. Personal experience suggests strict reliance on fitted N95 masks, gloves, and eye protection, even during quick sampling or transfer. Laboratory safety data sheets spell out these risks, and they advocate robust local ventilation and frequent equipment cleaning. Training sessions for new team members always underline the importance of keeping acids and bases separate during storage, since mixing—even in small quantities—triggers exothermic reactions or splashes. Over decades of research, regulatory agencies like OSHA and the European Chemicals Agency have updated their hazard ratings, but responsible users still prioritize local SOPs. Waste streams containing Molybdenum Hydroxide demand labeling and careful neutralization before discharge, protecting both workers and the wider environment.
Application Area
Industries worldwide look to Molybdenum Hydroxide to solve practical challenges. The compound steps up as a solid catalyst in oxidation and hydrogenation reactions. Paint producers harness its mild coloring ability and corrosion resistance for specialty pigment formulations. Oil refiners depend on molybdenum-based catalysts—including the hydroxide form—to drive desulfurization processes, cleaning up petroleum products for cleaner combustion. In electronic material manufacturing, thin films containing molybdenum offer unique electrical and thermal properties, finding spots in semiconductor devices and sensors. On the research side, innovative minds exploit the compound’s redox capabilities to tackle water purification, battery development, and new routes to biodegradable plastics. My personal work with environmental applications uncovered its talent for breaking down persistent pollutants in wastewater, proving that even laboratory-scale experimentation can bring relevant solutions to industrial-scale problems.
Research & Development
Laboratories around the world push boundaries in the chemistry of Molybdenum Hydroxide. Teams at leading universities dig into its fundamental structure using high-resolution X-ray crystallography and neutron diffraction, searching for interlayer arrangements that promise higher catalytic outputs. Researchers in the sustainable energy sector keep testing it for next-generation batteries and hydrogen production technologies. Funding agencies encourage work on modification techniques—such as doping, exfoliation, or assembly into mesoporous materials—that could change energy storage or water-splitting efficiency overnight. Start-ups and major corporations alike invest in pilot projects where molybdenum hydroxide catalysts slash emissions or boost chemical yields. Results from these R&D efforts point to real-world impact over the coming years, proving the value of close collaboration between public funding, academia, and industry.
Toxicity Research
Careful readers of toxicology reports already know that Molybdenum Hydroxide, like many inorganic compounds, poses manageable but serious risks. Direct ingestion by lab animals at high doses brings out signs of toxicity, such as gastrointestinal irritation and transient changes in enzymatic systems involved with copper metabolism. Occupational exposure studies over several decades highlight that airborne particles at excessive concentrations raise risks of lung inflammation and potential long-term effects on liver and kidney functions. Regulatory limits—set by global agencies—reflect these findings and make certain workplace air stays clean and that wastewater outflows fit environmental standards. Experience in industrial labs confirms that routine monitoring, proper training, and up-to-date emergency procedures effectively keep incidents rare, even as worldwide production and application increase year by year.
Future Prospects
With growing demand for efficient catalytic materials, energy storage devices, and advanced sensors, Molybdenum Hydroxide stands out as a likely cornerstone in future industrial and scientific breakthroughs. Expansion of hydrogen economy projects calls for ever-better catalysts, often drawing on precisely engineered molybdenum compounds. Environmental imperatives push industries toward materials that combine performance with reduced toxicity and easy recyclability, which encourages even more research into custom-tailored modifications. Ongoing projects in green chemistry departments aim to unlock improved synthesis routes that cut down chemical waste. Several start-ups now partner with major chemical firms, betting that molybdenum compounds will supply solutions that support the transition to cleaner technologies without eroding profit margins. Over my career, watching research projects grow from small-scale benchtop work to pilot-scale production testifies to the untapped opportunities still present in the field, where theory, regulatory frameworks, and real-world needs join forces.
A Look at a Quiet Performer
Many people pass through their workdays without thinking about the strange and rare materials running the show in the background. Molybdenum hydroxide rarely makes headlines or pops up in conversation, yet this compound touches several fields most people would never expect. If you poke around in labs, refineries, or even places where energy tech gets built, you might notice its signature blue-green hue dotting workbenches and notebooks.
Catalysts Changing Chemistry
In chemical plants, people rely on molybdenum hydroxide when tweaking reactions or cleaning up industrial leftovers. Its main strength: it plays a big part in catalysis. Beyond textbooks, the reality is that making fuels or even basic plastics often needs a gentle and reliable push from a reliable helper. Molybdenum hydroxide sometimes acts as the starting material in the synthesis of more advanced catalysts that speed up difficult reactions without falling apart under pressure. Refineries often depend on catalysts derived from these molybdenum compounds to break down crude oil, helping squeeze out as much usable product as possible. Cutting corners on catalysts usually results in lower-quality fuel, extra waste, and more emissions.
Batteries and Energy Storage
The energy business always craves new ways to pack in more electricity and keep it from leaking away. Researchers over the past decade have experimented with molybdenum hydroxide in battery electrodes, including lithium-ion, sodium-ion, and supercapacitors found in electric cars and grid projects. Lab studies have shown that this compound can help boost charge times and hold onto energy for longer, especially when combined with other metals or nanotech. Some of these studies link its unique layered structure to better performance compared to more traditional electrode materials. In places looking to ditch fossil fuels, every slight edge in battery tech matters a lot.
The Role in Corrosion Protection and Pigments
Factories and engineers don’t often shout about corrosion, but rusted metal quietly drains money and effort. Molybdenum hydroxide sometimes goes into protective coatings for steel and iron. Its ability to resist chemical breakdown helps paints last longer on bridges, ships, or machinery exposed to harsh weather or chemicals. Over time, coatings built with this kind of ingredient help slow down maintenance cycles and cut cleanup costs. Besides protection, some artists and companies also find value in its color. Its pigment properties give certain ceramics and commercial paints that blue-green edge when designers want something eye-catching and durable.
Bridging Academic Curiosity and Real-World Needs
My own work in materials research exposed me to all sorts of clever solutions for common problems. One thing became clear: raw materials like molybdenum hydroxide make a difference where precision counts. Small breakthroughs—better catalysts that slash factory waste, or battery tweaks that push cars further—often start with old compounds used in new ways. As demand grows for greener tech, cheaper processes, and reliable infrastructure, people working behind the scenes rely on these hidden helpers. Molybdenum hydroxide won’t ever become a household name, but people counting on smoother commutes, cleaner air, and more affordable energy benefit from the quiet work it does.
The Science Behind Molybdenum Hydroxide
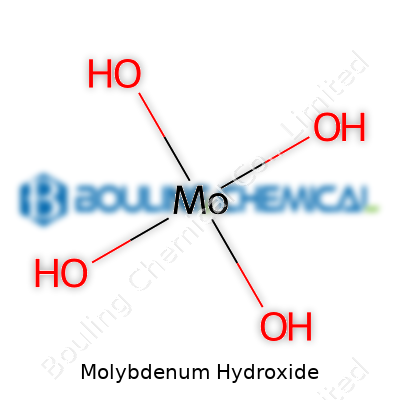
Molybdenum hydroxide appears in chemistry classes early on as a practical example of transition metal compounds. The chemical formula for molybdenum hydroxide is Mo(OH)3 for the common trihydroxide form, though a dihydroxide form, MoO2(OH)2, also exists. Most educational and research references talk about Mo(OH)3, so that is usually the main focus in discussions about this compound. This formula shows that a molybdenum atom connects to three hydroxide groups.
Personal Take: Why Molybdenum Hydroxide Matters
A lot of people hardly think about molybdenum outside industrial circles or fertilizer discussions. The exposure I’ve had working with chemical educators taught me that teaching students about rare but important elements pays off, mostly because they reveal patterns that run across the periodic table. Molybdenum sits in the middle of topics ranging from steel alloys to micronutrient cycles in agriculture. That same versatility comes up when chemists use molybdenum hydroxide as a starting point for creating catalysts or other molybdenum-based chemicals.
There’s practicality here too. Some industry specialists use molybdenum hydroxide as an intermediate for producing molybdenum oxide, a catalyst in petroleum refining. One senior chemist I once worked with drilled home the usefulness of hydroxides as precursors. They react cleanly and handle shifts in oxidation states gracefully, which simplifies steps in manufacturing processes.
Bridging Science and Industry
Stepping away from the classroom, the real-world use cases for molybdenum hydroxide start to reflect global shifts. Many modern technologies, such as hydrodesulfurization, need clean and effective catalysts. Molybdenum compounds answer that call, especially in processes aiming to get sulfur out of fuels. The refining industry measures catalyst success through both efficiency and longevity. Molybdenum hydroxide, transforming into oxides at elevated temperatures, helps manufacturers boost performance while keeping costs in check.
Another interesting point—some research shows that certain molybdenum hydroxide variants could become part of future battery or energy storage technologies. As the world leans away from fossil fuels, interest in these overlooked substances surges among energy researchers.
Concerns and Solutions
One nagging issue stands out: molybdenum compounds must be handled properly. Their environmental footprint depends on disposal methods and the possibility of leaching into water systems. Runoff or poor waste management can trigger toxicity concerns, especially for aquatic life. Experience working on lab safety committees taught me that strict protocols make a difference. Labeling, containment, and waste neutralization stop accidents before they start.
Industry and research labs can do better by setting tighter controls on effluent and pushing for greener synthesis methods. Some new techniques cut out harsh byproducts, swapping them with water-based processes. These solutions require steady investment and regulatory incentives, but history shows progress follows increased attention to health and environmental risks.
Looking Ahead
As education, industry, and environmental stewardship increasingly intersect, it’s worth recognizing the role of substances like molybdenum hydroxide. The formula may look simple: Mo(OH)3, but its applications touch on innovation, global industry, and sustainability. New advances and conversations in chemistry should keep expanding on safer, smarter ways to use these compounds. People sometimes forget that deep understanding starts with basic facts and careful use, both in the lab and out in the world.
Understanding Molybdenum Hydroxide in Everyday Context
Molybdenum hydroxide sounds like one of those chemicals tucked away deep in an industrial manual, but it pops up more often than most realize. Most folks might not recognize it by name, but it’s a compound with growing demand in research, electronics, and some specialized industrial roles. With more interest comes more questions: does it pose a hazard, and just how risky is it to be around?
Fact-Checking Toxicity Concerns
Molybdenum itself is a trace element needed by human bodies, mostly in tiny amounts. It helps enzymes fire up some important reactions, especially when breaking down proteins. Problems only pop up when people breathe, ingest, or come into skin contact with certain chemical forms of molybdenum in larger amounts. But everyone wants to know if molybdenum hydroxide belongs on the list of sketchy compounds that require extra caution or if it’s more benign like table salt.
According to reliable chemical safety databases and peer-reviewed research, toxicity from brief or casual exposure to molybdenum hydroxide isn’t something that should keep folks up at night. Animal studies show that very high doses over long periods can trigger symptoms — joint pain, fatigue, and in rare cases, gout because of how the compound interacts with copper in the body. It takes a lot, though. The amounts most people see are nowhere near those levels. Still, standing over open containers with this substance all day in a lab is not a smart move.
Handling It in the Workplace
Some manufacturing jobs or jobs in research might create more regular contact with molybdenum hydroxide powder. Here, the story shifts a bit. Like a lot of powdered metals and rare chemical compounds, it can irritate the eyes, the nose, or the throat if care gets tossed aside. It can also become a dust hazard if basic safety rules get skipped. Respiratory masks, gloves, and basic ventilation aren’t just suggestions for people working with this kind of material — they are common-sense tools. OSHA and European chemical safety groups back up those steps.
This approach isn’t fear-mongering; it’s about simple prevention. A bit of molybdenum hydroxide dust won’t send someone to the ER, but repeat exposure, especially in closed-off spaces, deserves respect. Company training, labels in plain language, and everyday safety gear go a long way to keeping problems rare. If folks report odd symptoms or chronic exposure, periodic health checks are warranted to keep everyone safe and catch issues before they grow.
Disposal and Environmental Points
Molybdenum hydroxide won’t rot away like a banana peel. It also doesn’t rank next to mercury or lead for long-term risk. Dumping solutions or large quantities straight into drains or ordinary landfills isn’t smart, since it might leach into groundwater. Local and national disposal guidelines exist for a reason — following them means fewer headaches for communities and cleaner soil and water in the long run.
With clearer knowledge, workers and regular folks alike can stay safe and avoid turning practical compounds into real hazards. Sound science, straight talk, and everyday precautions are the way forward.
Lessons Learned About Storage
Storing molybdenum hydroxide can make or break safe lab practice. This material doesn’t just get by on its own. It reacts to moisture and temperature shifts in a way you notice the hard way if you skip the basics. I found out early on that keeping the container tightly sealed makes the biggest difference. Any humidity creeping into the jar starts causing clumping, while the surface sometimes turns pasty—nobody wants to scoop out sticky powder for analysis.
Shelving options matter more than you’d guess. Store this chemical in a dry spot, away from the window where temperature fluctuates all day, and far from benches where acids get used. If it stays in a low-humidity cabinet away from direct light and vibration, your bottle lasts longer and the contents work as expected. Tossing a desiccant packet into the storage box never hurts, either.
Handling Brings Its Own Hazards
Lab gloves save you from skin exposures, but it’s about more than just wearing one pair. I’ve seen accidents happen when someone hurries and skips eye protection or forgets to wipe down the bench. Powders get everywhere once the seal breaks, and with molybdenum hydroxide, a little carelessness means it can sneak into the air or onto your hands. It shouldn’t surprise anyone—most forms of this metal compound should not be inhaled or absorbed through the skin.
Some researchers use automated weighing stations or fume hoods for every transfer, and while not every lab can afford these, working close to an extraction fan is important. Sweep up any spills while they’re fresh, and don’t try to reuse swept-up powder. The less this compound gets disturbed, the less likely you breathe it in.
Why Storage and Handling Advice Matters
People in the field remember moments when shortcuts led to issues: impurities in test results, surprise reactions when combined with acids, or unforeseen corrosion on shelving. Handling errors with molybdenum hydroxide can mean lost time on investigations, wasted materials, or even a visit to occupational health.
Medical journals point to chronic inhalation risks, highlighting compounds like these as potential causes of lung and kidney irritation. That knowledge changes your approach in every step. It’s not just about following a list of rules, but about understanding what small oversights do in real practice. Wear a dust mask for even short transfers, especially if you don’t have a downdraft table. This isn’t just bureaucratic safety—most of us have worked through coughing fits that ended up being avoidable.
Solutions That Actually Work
Portable dehumidifiers around storage shelves, locked bins, and clear SOPs drafted by the people who actually use the chemical benefit everyone. Double containers offer a second barrier against spills, and labeling bottles with the date opened helps flag staleness before performance drops. Custodians or lab members should swap out old stock and inspect seals regularly, since a little bit of prevention holds off a lot of headaches.
Training goes further when people with hands-on experience teach it. The person who scrapes crusted powder from lids or cleans up spills knows more than the sign on the wall. Bringing those war stories to new staff ensures they respect the risks and understand why the rules make sense—it's never just for show.
Listening to feedback on safety measures in your workspace can shape smarter routines and keep everyone healthy. Investment in better equipment pays itself back in fewer sick days, less spoiled material, and a more sustainable operation over the long haul. This attitude not only protects individual workers but forms the backbone of trustworthy science.
A Metal That Keeps Systems Running
In my years following the ins and outs of industrial chemistry, few specialty chemicals have kept so many systems humming as molybdenum hydroxide. Most folks don’t realize how often they rely on a small handful of compounds, but head into a metal refinery or a chemical plant, and you’ll quickly see its fingerprints everywhere.
Essential to Tougher Steel
Workers in the steel world depend on additives that stand up to stress, heat, and rust. Molybdenum hydroxide finds its way into steelmaking for exactly these reasons. High-strength steels use molybdenum-based compounds, which push back against corrosion and help the metal hold together under pressure. Builders, automakers, and bridge engineers count on these improvements; the safety and lifespan of the products they make get a lift because molybdenum brings real-world strength.
Catalysts for Cleaner Chemistry
Talk to anyone developing catalysts, and molybdenum comes up early in the conversation. Refineries and chemical manufacturers use molybdenum hydroxide to refine fuels, crack hydrocarbons, or drive other key reactions. Over decades, it’s helped the chemical industry make processes more efficient and a bit greener, by raising output and cutting waste. Instead of using more hazardous or expensive materials, many teams trust the reliable push that molybdenum hydroxide provides, especially in processes like desulfurization that pull sulfur from fuels—critical if you care about smog and clean air.
Pigments with Staying Power
Paint makers and plastics producers have come to value colorants that don’t fade, rust, or fall apart with time. Molybdenum-based pigments have built a reputation for making reds, oranges, and yellows that stick around, even on outdoor equipment and signage. With so many products needing to survive sunlight, weather, or chemicals, manufacturers aren’t just painting for looks—they’re painting for long-term durability. In my own experience, companies big and small have leaned on molybdenum pigments to save on touch-ups and stand up to abuse.
Electronics and Battery Tech
Newer applications keep cropping up, and battery researchers and electronics builders are especially interested. Molybdenum hydroxide helps in fabricating electrodes, especially as the tech world experiments with next-gen batteries and supercapacitors. Research papers stack up on my desk every year, each hinting at tweaks in performance, safety, or recharge time, and quite a few point toward molybdenum compounds as part of the solution.
Looking Toward Safer and Greener Practices
The appetite for safer, more sustainable processes grows each year. Choosing the right additive or catalyst matters. While regulatory pressure nudges some companies away from toxic or heavy-metal-based chemicals, molybdenum compounds, when handled with respect, fit into processes with less risk than some rivals. Still, it’s not a free pass—manufacturers continue searching for ways to recycle, recover, or safely dispose of waste.
Challenges and Solutions Ahead
One challenge comes from sourcing. Molybdenum, like many metals, depends on good mining and recycling networks. Sometimes prices swing, and industries have to hustle to manage costs. Researchers, myself included, push for better recycling and recovery, especially from used catalysts and spent industrial products. Smarter process design and tighter regulations can stretch supplies and keep demand in check.
In the end, the story of molybdenum hydroxide mirrors so many key industrial trends: making things stronger, cleaner, and more durable, without losing sight of safety or cost. Those of us watching the field know its uses aren’t going away soon—but we’re already chasing the next innovations that keep industry moving.