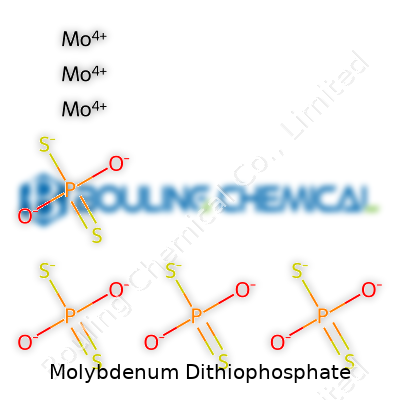
Molybdenum Dithiophosphate: A Closer Look
Historical Development
Molybdenum dithiophosphate entered the conversation among chemists trying to develop stronger, longer-lasting lubricants back in the 20th century. As cars multiplied and engine loads increased, the hunt began for additives that could do more than zinc dialkyldithiophosphate (ZDDP). Researchers in the postwar industrial boom found that combining molybdenum with phosphorus and sulfur unlocked new possibilities for reducing wear and friction under punishing conditions. Lab trials started modestly; a few grams here and there in small batches of oil. By the 1970s, as environmental regulations tightened and engines grew smaller and hotter, automotive and machinery manufacturers started asking whether these lab stars could be produced at scale, given the clear results: less wear on parts, fewer breakdowns, and extended life. Early patents and technical papers from Europe, North America, and Japan quickly shaped an industry eager for an edge. Today’s use of molybdenum dithiophosphate springs out of those madcap years when the oil crisis and pollution debates accelerated innovation.
Product Overview
Molybdenum dithiophosphate offers much more than basic lubrication. It typically appears in liquid or sometimes semi-solid form, dark in color and with a distinct odor that hints at its sulfurous chemistry. You will find it blended into engine oils, industrial greases, and even hydraulic fluids. This compound doesn’t work solo – oil formulators pair it with conventional base stocks and other performance boosters to cut down wear, keep metal surfaces separated under load, and tackle corrosion. The demand for cleaner, higher-performance lubricants in both automotive and industrial sectors keeps this chemical relevant, especially where old ZDDP alone can’t tackle modern challenges. Machinery running at higher speed and temperature sees benefit right in the field, not just in the lab.
Physical & Chemical Properties
Handling molybdenum dithiophosphate, you notice the color first—often deep amber or brown, sometimes almost black, with a heavy, somewhat pungent aroma. It dissolves into a range of mineral and synthetic oils, remaining stable through wide swings in temperature. The compound features both molybdenum and phosphorus atoms, joined with sulfur, a trio that reacts dynamically under pressure and heat. It resists breakdown at high temperatures, holding its own above 200°C in service. Chemists see a robust molecular structure here—one reason for its resistance to oxidation and its storage life in well-sealed drums or tanks. In ordinary conditions, it stays fluid but gets viscous at lower temperatures, enough to make careful handling necessary during winter.
Technical Specifications & Labeling
Industry protocols demand transparent labeling of phosphorus, molybdenum, and sulfur content by weight, often detailed to tenths of a percent. Oil marketers provide data sheets that lay out viscosity at 40°C and 100°C, acid value, saponification value, and flash point—numbers that buyers in engine assembly or heavy machinery rely on. Most suppliers print batch numbers and recommended handling temperatures on barrels. Service shops notice “MoDTP” on spec sheets or lubricant packaging, along with compatibility statements for popular base oils and seal materials. Some global regulatory agencies set caps on phosphorus and sulfur to prevent catalyst poisoning, so compliance documentation travels with every shipment.
Preparation Method
Chemists preparing molybdenum dithiophosphate usually start by reacting molybdenum compounds such as molybdenum oxide or ammonium molybdate with dialkyl or diaryl dithiophosphoric acid under controlled temperatures. The process releases water or ammonia as byproducts. Getting the proportions right determines the final additive’s performance, so laboratory scale-ups demand precision with mixing, heating, and distillation. Modern plants use stainless steel reactors, inert gas blankets, and closed-loop systems to capture and treat sulfur-rich fumes. Quality control labs sample batches for both active molybdenum level and hydrolytic stability before anything ships for blending.
Chemical Reactions & Modifications
Engineers in oil labs know that molybdenum dithiophosphate transforms once the engine fires up. Under heat and pressure, the compound decomposes at metal surfaces, forming films rich in molybdenum disulfide and iron phosphate. These slippery layers adhere right where metal meets metal–cams, lifters, bearings, and gears. The decomposition also throws off phosphoric acid derivatives that buffer the onslaught of combustion byproducts. Tweaks to the alkyl group – for example, swapping out butyl for octyl – can alter solubility and film thickness, letting chemists fine-tune performance for different engines or industrial gearboxes. Acid and base catalysis both promise subtle ways to shift the compound’s action, driving continuing research.
Synonyms & Product Names
Look at spec sheets worldwide and you see molybdenum dithiophosphate sold under several names. Common synonyms include MoDTP and molybdenum O,O-dialkyldithiophosphate. Commercial labels often reflect the composition, such as Moly Add 4080 or Seamol 1050. Some suppliers emphasize regional naming conventions; in Asia, you might see trade names carrying random numbers but always referencing the molybdenum and dithiophosphate roots. Anyone buying or blending must check the product data sheet for CAS number confirmation to avoid costly mix-ups.
Safety & Operational Standards
Plant workers don chemical goggles and gloves before opening a drum of this material. Inhalation of vapor or prolonged skin contact invites irritation, so personal protective equipment is non-negotiable. Storage calls for cool, dry, well-ventilated facilities, away from strong acids or oxidizers. Local fire codes often require spill containment because of the risk of sulfur-laden runoff in an accident. Regulatory authorities in Europe and North America publish exposure limits and call for environmental monitoring, since improper disposal threatens soil and groundwater. Emergency wash stations stand close to storage areas. Transport moves under hazardous material rules, with UN numbers and clear hazard statements written on shipping containers.
Application Area
Automotive engine oils carry the largest demand, especially for high-performance cars and heavy trucks where engineers chase every percentage point of efficiency. Molybdenum dithiophosphate earns its keep inside racing engines, off-road machinery, and industrial compressors running days or even weeks between shutdowns. Besides engines, you see this additive in metalworking fluids, greases for mining, and hydraulic oils facing punishing loads. In Asia, rapid urbanization feeds extra demand for construction equipment lubricated with high-moly blends. Wind turbine gearboxes in Europe benefit from the anti-scuff and load-carrying properties, locking in reliability through winters and storms. Factory maintenance teams count on fewer surprise repairs and pit stops, extending asset life while controlling costs.
Research & Development
Universities and private labs run an arms race against friction and wear, testing new versions with different alkyl chains, metal ratios, or even hybrid molecules that blend molybdenum with tungsten or vanadium. Real-world engine tests, bench-rig friction meters, and high-resolution surface analysis reveal where today’s molybdenum dithiophosphate still falls short: phosphorus residue can damage emission catalysts, or the film thickness drops off after a few thousand miles. Researchers in Japan and Germany invest in nano-scale additives, chasing longer life, cleaner exhaust, and reduced greenhouse gas emissions. Patent filings map out fresh techniques for synthesis, purification, and blending. Industry-academic partnerships get bankrolled by automakers and lubrication giants looking for cleaner, more robust ways to protect metal.
Toxicity Research
Toxicologists assess molybdenum dithiophosphate on multiple fronts: acute and chronic exposure, ecological fate, breakdown byproducts, and persistence in groundwater or soil. Animal studies highlight irritation potential at high exposure levels, but don’t depict widespread systemic effects from incidental skin contact. Inhalation of aerosols during blending or application, though, raises concern given the sulfur and phosphorus content. Regulatory pressure in the EU and China pushes for lower allowable concentrations in finished oils. Waste handling and recycling policies in North America watch for trace contamination in spent oil streams and used oil filters. Labs continue to probe for any subtle health effects tied to long-term, low-level exposure among technicians and plant workers. Limited data exists on bioaccumulation, but careful monitoring remains part of any responsible safety program.
Future Prospects
Shrinking pollution limits and tougher engine standards leave room for innovation. Automakers want lubricants that cut friction without clogging emission controls. Molybdenum dithiophosphate will stick around as long as internal combustion engines and heavy machines roll off assembly lines, but pressure grows for greener, phosphate-free options that still fight wear reliably. Nanotechnology and green chemistry approaches beckon, from bimetallic complexes to biodegradable alternatives. Digital modeling tools help chemists imagine new molecules and test failure points in silico, speeding the process toward safer, longer-lasting lubricants. The market moves forward on both ends: meeting strict environmental standards and squeezing ever more efficiency from every drop of oil. As electric vehicles enter the mainstream, researchers will need to push past old assumptions, asking if this time-tested chemical can morph into the next essential additive or yield ground to new, cleaner molecules. For now, maintenance managers, plant operators, and gearheads still count on molybdenum dithiophosphate to keep engines running strong.
The Backbone of Modern Engine Oils
Every time an engine starts, thousands of metal parts begin to grind, slide, or push against each other. Without solid protection, moving parts won’t last very long. Over the years, I’ve learned the hard way: cars rely on more than just oil—additives play a huge part. Molybdenum dithiophosphate, given its long name, sounds complicated. The job it does isn’t. This chemical helps stop metal surfaces from welding together under high pressure and heat. It forms a thin film, so parts glide instead of clash. Less friction means cooler engines, quieter rides, and fewer breakdowns.
Why Lubricants Get the Spotlight
Wear and tear doesn’t discriminate. Tiny gearboxes in an assembly line or turbocharged engines feel the same pressure to stay reliable. Under heat and the everyday stress of driving, oil alone can’t quite cut it. There’s sulfur, zinc, phosphorus, and, yes, molybdenum compounds. When everyday folks drop their car for an oil change, most don’t realize that the right additives save us all from hefty repairs. I’ve met truck drivers and engineers who swear by high-quality oil, and the benefits always circle back to a cocktail of additives, with molybdenum dithiophosphate standing out for its anti-wear reputation.
Fighting Friction and Corrosion
You can spot the difference between engines maintained with good oils and those that go cheap. Fewer deposits build up, and metal parts show less scoring. Molybdenum dithiophosphate steps in here. It forms a strong protective layer on hot metal surfaces, slashing friction and holding off corrosion. High-stress driving, stop-and-go city traffic, and heavy loads punish engines daily. This protective film, born from a mix of phosphates and molybdenum, gives parts a fighting chance. The research backs up these stories. Tests often point to 60% lower wear when this additive is in play compared to bare oil.
Today's Challenge: Environment and Efficiency
Emissions standards keep tightening. Car manufacturers know every small win matters for both pollution and fuel efficiency. Molybdenum dithiophosphate doesn’t just protect—it helps fuel burn smoother, trims emissions, and stretches out the engine’s life. Cleaner oil, fewer ash-forming elements, and better combustion all tie back to this additive.
Looking for Safer and Smarter Alternatives
Every solution brings new challenges. There’s growing pressure to limit phosphorus and sulfur additives in oil, since these elements can harm catalytic converters in modern exhaust systems. Oil makers want alternatives that protect engines just as well, but don’t leave behind harmful traces. Researchers across the globe explore changes to the molecular structure of molybdenum dithiophosphate, hoping to keep the good—like anti-wear and friction-busting—and drop what hurts air quality. Water-based lubricants stand out on the horizon, and nanotechnology brings new hope for smarter, cleaner lubrication.
Choices That Matter
When picking engine oil, choosing the right additives means fewer headaches down the road. For truckers, commuters, or factory floor supervisors, smarter lubrication helps keep wheels—and industries—moving. From what I’ve seen, the humble molybdenum dithiophosphate deserves a little respect, quietly working to cut friction, save energy, and keep engines running well beyond their warranty.
The Real Story Behind Engine Additives
Every time I pop the hood on my aging sedan, I’m reminded how far engine oil technology has come. Additives do a lot of heavy lifting when it comes to modern engine protection. Molybdenum dithiophosphate (MoDTP) is one name that shows up on a lot of lubricant spec sheets. Designed for improving wear resistance in oil, it’s easy to overlook the safety side of things while focusing on all that performance hype. But nifty chemistry in those bottles comes from a long line of rigorous research.
Shedding Light on Hazards
Chemists know that MoDTP has some bite to it. We’re not dealing with table salt or flour here. Left on the skin, the compound can cause irritation or redness. Let it splash into the eyes, and pain follows. Long-term exposure — especially among folks who handle raw materials or pour additives without proper gloves — may set them up for dermatitis or worse. Inhaling dust or fumes from heated MoDTP breaks peace at the lungs and throat too.
MoDTP doesn’t release fumes at room temperature in a home garage, but the risks jump once it’s processed or heated in industrial settings. Chronic overexposure in factory environments has the potential to impact the kidneys or liver. Most folks will never face that, but this isn’t a risk worth taking lightly. Oil blenders and plant technicians must pay attention far more than your average motorhead.
How Factories and Professionals Avoid Trouble
Serious oil plants lock away MoDTP in steel drums in well-ventilated storage rooms, far from food or living things. Chemical gloves, splash goggles, and lab coats are more than just a suggestion in these places. Smart technicians lean on exhaust fans or fume hoods during mixing, not just to follow guidelines, but to dodge headaches and chemical coughs.
Thorough cleanup after a spill means using absorbent materials and then tossing them as hazardous waste — not just sweeping it under a rug. Good engineering controls and a strict cleanup routine make all the difference. One real-world story: I saw a workplace skip proper training, and soon after they faced a rash of skin complaints. It only took a couple afternoons of solid safety orientation for those complaints to drop off the radar.
For DIY Mechanics and Everyday Users
In my own garage, engine oil top-offs happen with gloves on. I keep rags handy and wash up before grabbing a sandwich. For anyone mixing their own additives, safety goggles join the party. It’s easy to forget these basics on a Saturday afternoon, but itchy skin isn’t a badge of honor. One missed glove and an unexpected splash can ruin your day.
MoDTP shows up in finished engine oils at safe concentrations. The real danger lurks during handling of raw, concentrated forms. At home, stick to the intended uses — never try blending your own without understanding the hazards. Respect for chemicals is as important as torque specs and oil weights.
Building a Safer Oil Culture
Trustworthy brands include hazard information right on the Safety Data Sheet. This bit of bureaucracy can be a lifesaver for those paying attention. Local waste regulations often ask for used oil to be properly labeled and recycled — don’t dump chemicals just because out of sight means out of mind. Shared stories and tips among gearheads make the rounds, but a careful eye for safety keeps cars and people running longer.
Performance Under Pressure
Engines and machines run harder than ever, and friction isn’t just a word to engineers—a little bit can chew through metal over time. Molybdenum dithiophosphate (MoDTP) steps into that fight. It doesn’t just slow wear; it changes how steel surfaces handle punishment. A thin, persistent layer forms between moving parts, shielding them from grinding and damage. Real-world testing proves it: motors with MoDTP in the oil rack up more operating hours before anyone spots scoring or scuffing on surfaces.
I’ve worked around heavy machinery. You can’t miss the difference in tear-downs after switching to a molybdenum additive. Gears that looked beat up after a season suddenly wear down much slower. Some shops swear by it for high-load applications: forklifts, mining engines, or anything that runs hot all week.
Fighting Corrosive Challenges
Engines face another enemy beyond friction—acidic byproducts from fuel or contaminants try to eat away at metal. Molybdenum dithiophosphate steps up, especially where conditions get nasty. The additive fills the gap left by older, chlorine-heavy solutions that brought environmental headaches. Doing the job without adding new pollution risk matters for any shop that plans to last.
I’ve seen what acid corrosion looks like in gearboxes—deep pitting that no oil alone could prevent. Fluids with MoDTP noticeably slow the attack, even in systems exposed to condensation and dust. Industrial bearings that used to pit out after a wet winter survive much longer, and that saves hassle and money.
Boosting Oil Life and Engine Cleanliness
Additives must do more than reduce wear; the oil itself needs longer life if engines are going to keep working without constant filter changes. MoDTP’s antioxidant punch makes it harder for heat and oxygen to break oil down. That means less sludge in hard-worked engines and fewer oil changes for vehicles that clock serious mileage.
In my own fleet days, long-haul rigs lasted well beyond factory recommendations between oil swaps once MoDTP became a staple. Instead of thick black sludge on drain day, used oil often came out in better shape—still golden, still slick. That kind of result cuts downtime and stretches a maintenance budget further.
The Environmental Factor
There’s an ongoing shift to get heavy metals and hazardous chemicals out of lubricant packages. Molybdenum’s profile looks pretty appealing compared to old-school options that relied on zinc or chlorine compounds. Emissions testing and wastewater challenges keep getting tighter, so shops and lube manufacturers pay close attention. MoDTP helps meet stricter rules without giving up performance—nobody wants to bounce between protecting engines and protecting the air or water.
Plenty of lab results show MoDTP handles high temperatures and pressure without spiking emissions or clogging after-treatment filters. That fits right alongside stricter global emissions regulations. Engines stay cleaner, and used fluids don’t create as much disposal trouble.
Solutions for Tomorrow’s Engines
Engines evolve, and with electric vehicles and hybrids picking up steam, lubrication faces new challenges. MoDTP continues to earn a place thanks to its proven toughness and its ability to play well with next-generation oils. R&D teams constantly look at the balance between performance, cost, and safe chemistry. MoDTP keeps showing up in the blend, and for good reason.
Shops and fleets looking for fewer breakdowns and longer service intervals have every reason to consider an oil package built around molybdenum innovation. The long-term view always favors tools proven in the field, especially under tough conditions—and so far, this is one additive with the track record to back it up.
The Realities of This Lubricant Additive
Molybdenum dithiophosphate (MoDTP) turns up in many lubricant blends because it helps machines live longer through its antiwear and antioxidant behaviors. In workshops and chemical stores, people see drums and pails of it stacked high. Some folks toss these containers in the corner and think the job is finished. A little hands-on experience proves careless handling doesn’t pay off later. Storing MoDTP the right way saves money, prevents hassle, and protects people and the environment.
Space, Temperature, and Stability
MoDTP does not take kindly to temperature swings or sunlight. From years spent in a plant, I have seen solidified or separated additives spoil full batches of lubricants. Store this material indoors at room temperature. Direct exposure to high heat, freezing, or sunlight causes clumping, sludge, or evaporation—none of which belong in a quality finished oil.
A thermometer on the storage wall is worth its cost many times over. I keep stacks of MoDTP at around 20°C (68°F). On days when things heat up, check for any swelling or leaks. Cold can thicken the product or change its chemistry. Move containers away from doors or drafty areas that hit with the changing seasons.
Picking Containers That Last
I learned this trick early: The original drums or jugs aren’t just for shipping—they protect the chemical from the air. MoDTP reacts with moisture or oxygen, so reseal every drum tightly after each use. Using clean, dry tools reduces the chance of introducing water or dirt into the mix.
Some plants like using dedicated, labeled storage bins or shelving. If old labels fall off or fade, confusion leads to trouble. Fresh, waterproof labels keep everybody certain about what’s where. Dangerous mix-ups can happen otherwise. Even new tech can’t beat a bold marker and clear signage when you want to store MoDTP safely.
Preventing Fires and Spills
It’s no secret that MoDTP belongs far from open flames or sparks. Most manufacturers highlight the product’s flash point, usually above 150°C (302°F), but careless ignition sources still spell disaster. Clear fire extinguishers, away from main walkways, and regular fire drills help staff act quickly if something goes wrong.
Spills turn slick fast. Concrete floors with a gentle slope toward a drain speed up cleanup. Store containers on trays that catch leaks, especially for large drums. This method stopped what could have been a major slip incident in my workshop—an ounce of prevention really does beat a pound of mop.
Handling Leftovers and Waste
Old or contaminated MoDTP cannot go in the trash. Most local rules demand disposal through certified hazardous waste programs. I set up a dedicated waste drum and track its fill level, alerting safety managers when it’s close to full. Posting the disposal steps with the drum eliminates questions.
Good Habits Save Money and Trouble
Proper storage of MoDTP reduces product loss, lowers costs, and protects everyone who handles it. Practice and simple routines keep sites safe. With clear labels, tight lids, indoor storage, and thoughtful disposal, storing MoDTP becomes one less thing to worry about while running a smooth operation.
Why This Chemical Matters
Molybdenum Dithiophosphate (MoDTP) stands out in the world of engine oils and lubricants. It helps keep engines running smooth by fighting wear and reducing friction. Drivers trust these blends to extend vehicle life and keep parts from grinding away too quickly. Yet as the focus moves from engines to ecosystems, the concern grows: What happens once MoDTP moves beyond the workshop?
Where Concerns Begin
Most people never see the chemicals flowing from engines into the environment. Even so, oil leaks out of cars, spills from service shops, or runs off highways every day. Storm drains grab these oily traces and send them toward rivers, fields, and lakes. Molybdenum Dithiophosphate does not just disappear once oil drips onto a driveway—chemicals linger, break down, or end up in soil and water.
Science shows that some parts of MoDTP, especially the phosphorus and sulfur pieces, cause trouble once they escape into water. Too much phosphorus fuels algae growth, choking ponds and starving fish of oxygen. Sulfur compounds can change soil chemistry, affecting crops or plant health. Molybdenum itself, though a trace mineral plants need, can harm animals in larger amounts. Livestock exposed to higher molybdenum in their water have suffered copper shortages, leading to illness or weaker growth.
What Research Says
Toxicity tests on MoDTP show mixed results. In small amounts, water creatures like fish seem to tolerate this chemical—regulators currently believe it breaks down and loses most of its punch. Problems build when used oil piles up year after year, or when filters and drained engine fluids pour into ditches unchecked. The complex structure of MoDTP makes it harder to trace in nature, and its byproducts have yet to be fully understood.
A 2021 assessment from the European Chemicals Agency handled this question head-on. They noted the lack of long-term research into how MoDTP builds up or breaks down in the environment. Scientists find it challenging to simulate years of runoff or oil spills in a lab, and even harder to measure the tiny amounts that collect season after season.
A Path Toward Safer Use
Auto shops and DIY mechanics face real choices every oil change. Collecting waste oil and sending it for safe disposal keeps most pollution out of the environment. City recycling programs take in used oil, and many shops will accept it back, making it less likely to soak into the ground. Communities that care for wetlands and waterways also work to stop stormwater from carrying oil off roads and into local streams.
Researchers continue searching for alternatives to chemicals like MoDTP—compounds that shield engines just as well, without causing as much worry after use. Some new additives promise to give the same protection, but cost or supply chain limits slow adoption. That trade-off between engine health and environmental safety shapes the lubricant market.
Balancing Engine Protection and Environmental Safety
People depend on cars and machinery, and engines depend on their chemical helpers. Yet the price for smooth pistons does not just show up in repair bills; it also surfaces in streams and soils. The challenge falls to manufacturers, mechanics, and anyone who changes oil—to find handling practices that protect both engines and the earth underneath them.