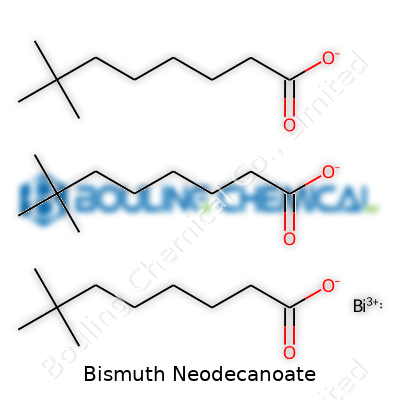
Bismuth Neodecanoate: From Discovery to Future Prospects
Historical Development
Chemists started paying attention to bismuth compounds long before neodecanoate ligands entered the stage. Bismuth itself sat in the toolkits of apothecaries and metallurgists centuries ago, mostly for its low toxicity compared to other heavy metals like lead. Fast-forward to the late 20th century, researchers chasing stable, oil-soluble metal salts landed on neodecanoic acid—a branched-chain carboxylic acid—which, when paired with bismuth, opened doors to new industrial applications. The shift came around the time regulators began frowning on lead and cadmium catalysts and driers, prompting the chemical industry to take bismuth seriously beyond its role in Pepto-Bismol and some alloys. As environmental rules toughened, teams from Germany to the US filed patents and explored processes for synthesizing and purifying bismuth neodecanoate that could scale and meet ever-tighter impurity limits.
Product Overview
Bismuth neodecanoate belongs to the organometallic family, specifically to the class of bismuth carboxylates. The commercial product shows up as a slightly viscous, pale-yellow to clear liquid, weighing in at roughly 25% bismuth by mass, the rest made up from neodecanoic acid residues. Most users pour it straight from metal drums or plastic jugs into blending tanks, focusing on its compatibility with a wide range of solvents and resins. Unlike lead, bismuth stays fairly benign in handling, a point not lost on formulators switching to less hazardous alternatives.
Physical & Chemical Properties
In practice, bismuth neodecanoate feels heavier on the hand than standard hydrocarbon oils. Its specific gravity hovers around 1.08–1.14 g/cm³, with a dynamic viscosity spanning 160–350 mPa·s at 20°C. It resists hydrolysis better than straightforward bismuth salts, surviving brief exposure to moisture without breaking down or separating. The neodecanoate backbone gives the molecule ample solubility in aliphatic and aromatic hydrocarbons, plus esters and glycols. Under a microscope or mass spec, the structure reveals bismuth bonded to three bulky neodecanoate ligands, which slows down unwanted side-reactions. In UV-cured coatings, this structure translates to a longer shelf-life and repeatable performance.
Technical Specifications & Labeling
Suppliers print bismuth content clearly—typically from 21% to 29%—based on the blend. Users should see acid values below 10 mg KOH/g, signaling decent purity and minimal free acid. Water content lands below 0.2% most of the time, and volatile organic impurities rarely exceed 0.5%. Color is measured by the Gardner scale, usually staying between 2 and 6. Modern safety data sheets spell out packaging volume, recommended storage, flash points (often above 110°C), and core hazard codes under GHS. Buyers watch for CAS numbers—the most cited is 34364-26-6—and UN numbers for transport, especially when shipping across borders.
Preparation Method
Bismuth neodecanoate starts with bismuth(III) oxide, one of the more stable bismuth salts, reacted with an excess of neodecanoic acid. This reaction is done at 120–150°C in a solvent like xylene, using nitrogen to prevent unwanted oxidation. The bismuth oxide dissolves slowly, making water as a byproduct, which is removed by distillation. Once the acid number falls to target range and the solution looks uniform, the mixture cools and gets filtered to catch any undissolved solids. Some plants tweak the ratio or strip out residual solvents, chasing better shelf life and performance in the end-use.
Chemical Reactions & Modifications
Bismuth neodecanoate steps in as a Lewis acid in urethane chemistry or crosslinking for alkyd paints. The molecule can transfer bismuth ions to reactive sites, gently boosting reaction rates without tearing up the molecular backbone of polymers. For more tweakable reactivity, chemists substitute a fraction of neodecanoate ligands with other carboxylate groups, tuning reactivity profile and solubility. Some R&D labs attempt ligand exchange to create hybrid metal soaps for custom catalysis, keeping process water and side-products in mind. Electrochemical methods rarely see use in the field but occasionally pop up in academic work testing new complexes for electronics or sensor use.
Synonyms & Product Names
Bismuth neodecanoate appears under a variety of labels in catalogs. Chemtrec and Sigma-Aldrich both list it as “Bismuth(III) neodecanoate” or “Bismuth tris(2,2-dimethyloctanoate).” Less often, the same product is called “Bismuth neodecanoic acid salt” or “Bismuth neononanoate,” reflecting regional naming customs or translation quirks. Brand-specific lines—from Shepherd to OMG—market it as a “bismuth drier” or “bismuth catalyst,” with spec sheets aligned to target sectors like coatings, adhesives, silicones, or elastomers. Proper documentation traces lot numbers from plant to barrel, reassuring downstream users about consistency.
Safety & Operational Standards
DuPont, BASF, and independent safety boards put bismuth neodecanoate through the usual gauntlet of toxicity and reactivity tests. It scores low on acute toxicity charts, especially compared to lead and cobalt salts, but safe practices apply. Users keep it off their skin, wear gloves, and avoid breathing in its vapors during transfer or mixing. Proper lab ventilation and labeled containers prevent most mishaps, while spill kits handle rare leaks or splashes. Waste bismuth residues end up in specialized chemical dumps or sometimes get recycled into new bismuth products, keeping heavy metals out of landfills. Fire-fighting plans note the high flash point and smoke hazards if overheated.
Application Area
Paint and coatings manufacturers count on bismuth neodecanoate for catalyzing the drying (crosslinking) step in alkyd and urethane systems. Automotive topcoats, furniture varnishes, and architectural finishes benefit from its effectiveness in humid rooms or under unpredictable temperatures. Silicone rubber companies deploy it in room-temperature vulcanization (RTV) catalysis, where its bismuth core replaces the old tin-based catalysts—another win for health and compliance managers. Polysulfide sealants, hot-melt adhesives, and newer 3D printing resins use similar bismuth chemistry. Increasing demand for phthalate- and lead-free products has pulled bismuth neodecanoate from a niche player to a mainstream additive in the past decade.
Research & Development
University groups and industry labs keep testing new blends of bismuth carboxylates, aiming for easy handling and higher activity. Some teams, especially in Japan and Germany, work on boosting metal loading or reducing the cost per kilogram without sacrificing quality. Others focus on finding smarter ligand structures that dissolve seamlessly in waterborne paints or recyclable polymer systems. Analytical chemistry breakthroughs—from nuclear magnetic resonance to high-res mass spectrometry—let scientists confirm product purity and stability in weeks, not months. Regulatory pressure to list every ingredient and residual trace element has made transparent supply chains a laboratory priority, shaking out unverified suppliers.
Toxicity Research
Bismuth neodecanoate earns praise for its low acute and chronic toxicity, but no chemical deserves blind trust. Chronic exposure studies on rats show less accumulation in organs than cadmium or antimony cousins. Skin absorption stays low, aligning with its oily, resinous nature. Nonetheless, agencies like the EPA and ECHA demand periodic reviews, focusing on inhalation risks and breakdown products during burning or incineration. Workplace exposure levels stay well below any threshold limit value (TLV) for career staff, though accidents or sloppy handling can still have consequences. Labs test for bismuth traces in process water and effluent, with many countries setting tight discharge rules.
Future Prospects
More manufacturers want metal catalysts with cleaner safety and environmental records. Bismuth neodecanoate steps toward that future, facing steady competition from modified zirconium, manganese, and rare-earth carboxylates. Market trends point to even stricter VOC and REACH standards in the EU, meaning the next wave of product tweaks will balance cost, solubility, and reaction speed even more tightly. Partnerships between universities and specialty chemical firms look to push ligand design and recycling schemes, aiming for circularity in a traditionally linear industry. As electronic devices get smaller and coatings more specialized, bismuth-based compounds, including neodecanoate, gently edge out legacy metals, delivering competitive performance without legacy hazards.
A Closer Look at What It Does
Bismuth neodecanoate pops up often in conversations about coatings and plastics, but its role stretches beyond just filling a formula sheet. In paint shops and industrial labs, workers depend on additives like this for the simple reason that oxygen and sunlight can mess with regular materials in ways we often overlook. Bismuth neodecanoate does the kind of work people often attribute to more traditional, and sometimes more toxic, compounds like lead. It speeds up drying, bumps up hardness, and helps keep finishes from feeling sticky long after application. Factories appreciate this chemical for its ability to make coatings behave how they’re supposed to, whether workers roll them out in big batches or small jars.
Why Use Bismuth Instead of Lead?
The conversation around lead has changed a lot due to health worries, especially in homes, toys, and workplaces. After reading dozens of studies and news reports, parents and regulators called for safer alternatives. Bismuth steps into that opening. The metal does much of what lead would but without the heavy baggage of neurotoxic risk and groundwater pollution. In practice, bismuth neodecanoate doesn't sacrifice the performance that industry expects, especially in urethane and alkyd paints, common on metal railings, pipelines, and machinery. It helps paints dry evenly and thoroughly. Unlike its more notorious cousins, this compound doesn’t carry the same long-term safety warnings.
Examining Its Reach Beyond Paints
Outside the world of coatings, bismuth neodecanoate finds a spot in plastics and rubber processing. Folks making PVC pipes or flexible tubing lean on stabilizers to deal with heat and prevent yellowing or breakdown. This additive stands out for helping keep materials sturdy, even when products live in direct sun or wild temperatures. I’ve met several engineers who prefer it in gaskets and seals that end up inside cars and appliances, where reliability matters more than sheer cost savings.
Regulation and Environmental Impact
Governments and agencies from Europe to North America follow bismuth-based compounds closely, and environmentalists track their adoption. So far, scientific reviews suggest it’s a safer option compared to older metal-based drying agents. In the production process, waste and runoff need management, but real-world risk remains much lower than with mercury or cadmium compounds. This helps factories meet tighter restrictions without constant dread of future liability. In a world where green chemistry shapes purchasing and policy choices, switching to bismuth neodecanoate can help manufacturers show leadership, not just compliance.
Where to Go from Here
Switching to bismuth-based solutions makes sense for manufacturers concerned about the future—not just their bottom line. They need to keep their products strong and safe, while buyers look for labels that promise fewer environmental headaches. More research always helps, especially as new uses pop up in electronics and batteries, but the track record so far lines up with healthy workplaces and cleaner communities. In my own work with industrial clients, those aiming for long-term credibility often lead the way by adopting compounds like bismuth neodecanoate before rules even require it. This is not just smart chemistry; it’s smart business too.
Chemical Formula and Structure
Bismuth Neodecanoate shows up quite a bit in industrial chemistry. The chemical formula is Bi(C10H19O2)3. That means you have one bismuth atom connected to three neodecanoate groups. The neodecanoate part comes from neodecanoic acid, which is a synthetic fatty acid. You might see it also written as bismuth tris(2,2-dimethyloctanoate), reflecting the branched nature of the neodecanoic acid component.
Why Bismuth Neodecanoate Matters
For years, the chemical world has looked for less toxic alternatives to lead and tin in various processes. Bismuth Neodecanoate offers a good solution, because bismuth generally shows less toxicity compared to lead compounds. Many manufacturers rely on this compound in the production of flexible foams, paints, and coatings—especially where you need a catalyst that won’t introduce unnecessary health risks. More demand for environmentally safer products pushes chemicals like this into the spotlight.
Applications and Benefits
In curing polyurethane foams, Bismuth Neodecanoate helps to control the speed and quality of the reaction. You probably lean on polyurethane in furniture, car seats, and insulation. Bismuth-based catalysts keep the process cleaner, steering clear of the issues seen with tin-based products, which can cause headaches over workplace safety and hazardous waste handling.
Outside foams, some printing inks and coatings rely on this bismuth compound. These industries face tighter restrictions on heavy metals, so a catalyst with a record of safer use stands out. As someone who pays attention to paint regulations and raw materials, a shift to bismuth-based chemistry makes a noticeable difference, both in compliance and public health impact.
Safety and Health Points
The chemical industry has spent time and money studying the toxicity of bismuth compounds. Bismuth Neodecanoate, thanks to the stable organic ligands, limits exposure to free bismuth ions, which helps keep risks lower than for other heavy metal catalysts. Factories pay close attention to ventilation and disposal, but risk assessments show lower environmental threats than alternatives with lead, mercury, or cadmium.
Challenges and Possible Solutions
Any shift to less toxic chemistry can bring cost pressures. Bismuth, though safer, doesn’t come as cheaply as lead. Supply chains for bismuth have some tight spots, particularly during global disruptions. To keep production steady, some companies start blending bismuth Neodecanoate with other low-toxicity co-catalysts, or they search for recycling programs to recover bismuth from post-industrial waste streams. Research into bio-based ligand alternatives could bring costs down if they match or exceed current performance.
Tighter industry standards around the world signal a lasting move away from traditional heavy metal catalysts. Experience shows that getting ahead of these changes, rather than waiting on regulations, rewards both reputation and bottom line. Bismuth Neodecanoate isn’t a silver bullet, but it moves the chemical sector in the right direction for both worker safety and the environment.
What People Need to Know
Bismuth neodecanoate, sometimes listed as bismuth(III) neodecanoate, pops up in paints, coatings, and some specialty plastics. Whenever a chemical like this finds its way into workplaces and products, questions start flying about safety. Folks who work around industrial chemicals every day want answers that don’t hide behind jargon or long-winded labels.
The Real Story About Toxicity
Looking at bismuth compounds, the reputation holds fairly steady: compared to toxic heavy metals like lead or cadmium, bismuth’s risks come in much lower. Medical workers might recognize bismuth from old-school stomach remedies. That history can lull people into thinking it’s always harmless, but this compound pairs bismuth with neodecanoic acid to form a metal-organic mix, and that’s worth a closer look.
In research so far, bismuth neodecanoate does not rise to the top of lists that flag hazards. Acute toxicity reports show only mild symptoms in most lab tests, often after large exposures not typical for most daily settings. For some workers, regular skin contact or inhaling vapors could pose a risk. At higher concentrations or over repeated exposure, mild skin irritation or digestive discomfort appears more common than dramatic or life-altering effects.
Comparing to Old Rivals
This compound replaces toxic heavy metals in many uses. For years, industries relied on lead and tin as stabilizers or catalysts. Regulators and safety managers have pressured manufacturers to swap those out for safer choices. Bismuth neodecanoate’s knack lies in reducing some of those dangers. It's been described as the “greener” alternative, and that label holds up in side-by-side studies—bismuth versions don't build up in the body the way lead does, and no links tie them to cancer in animal studies.
Keeping Health and Safety at the Center
Even if something rates lower on the hazard scale, complacency isn’t a good idea. The chemical still shouldn’t go down the drain or into the open environment. Workers need gloves, goggles, and good ventilation—simple moves that make a real difference. In my own experience handling industrial paints and catalysts, quick training and easy-to-understand safety sheets often worked better than stacks of technical paperwork. Real hazard control means treating every chemical with respect, bismuth or not.
Regulations and What We Still Need to Learn
No big restrictions land on bismuth neodecanoate in Europe or North America. Agencies flag it as low hazard but still recommend the usual limits for workplace air and skin exposure. That said, long-term studies on chronic exposure aren’t everywhere yet. Science sometimes forgets the folks on the factory floor. If you’re handling drums and tanks, you can’t always wait for every last detail to get published. Respecting emerging evidence and plugging gaps early helps head off the rare but real risk.
Looking Ahead
If safer alternatives come up, companies should stay open to switching over. Until then, taking good care with storage and handling will keep bismuth neodecanoate where it belongs—in products, not waterways or lungs. Everyday choices—using the right gloves, cleaning up spills, keeping good air flow—add up to a safer place to work and fewer worries at the end of the day.
Storing Chemicals Takes Real Know-How
Most people never think about what goes into storing specialty chemicals. Bismuth Neodecanoate isn’t your average household item. This compound finds its way into paint driers, catalysts, coatings, and even certain medical formulations. Even though it brings plenty of value to industrial processes, it demands respect in storage. Tossing it on a shelf just won’t cut it.
Sensitivity to Heat and Air
Long experience with chemical labs taught me that heat really does a number on many reagents—compounds like Bismuth Neodecanoate break down faster if left at high temperatures. Any site manager worth their salt checks the storage room thermometer. Heat speeds up unwanted reactions and sometimes even triggers dangerous situations. Most chemical distributors place this compound in a cool, shaded part of the warehouse, where direct sunlight can’t creep in.
Air exposure isn’t harmless either. If a container stays cracked open or the cap isn’t on tightly, moisture and air work their way inside. Over time, that can degrade the chemical or even cause a messy spill. Resealing and inspecting lids always pays off, even if it feels tedious.
Choose Containers with a Purpose
From years of sorting stockrooms, specialty chemicals were always put in their original, labeled containers. Bismuth Neodecanoate does best in tightly-sealed glass or HDPE plastic vessels. Metal often reacts with many organometallics, so glass or plastic wins out for safety and shelf life. Someone swapping out original packaging for a random bottle may save a few minutes but lose days to cleanup and reorder costs if something goes wrong.
Lock It Up and Label Clearly
Responsible storage means more than a safe spot and a good bottle. Bismuth Neodecanoate belongs behind locked doors, away from food, medicine, and daily supplies. It’s not something you want the janitor or a curious new hire to stumble across. The label tells the truth about what’s inside and gives emergency crews real, usable info if an accident ever happens. There’s no room for mystery in a good chemical storeroom.
Ventilation and Spill Plans Matter
Nobody expects an accident, but preparation saves lives. Any room holding this compound needs solid ventilation. Breathing in fumes from a spilled batch can irritate airways or cause more serious health effects, so a well-maintained exhaust fan is a must. Every site should run regular drills and check that spill kits sit within reach—not buried under clutter. The right absorbents, goggles, and gloves should be waiting before a single drop falls.
Stay Legal: Regulation Isn’t a Game
Regulations surrounding chemicals also come into play. OSHA and local fire marshals offer plenty of rules for a good reason. Keeping records on who handles what, how much sits on hand, and how it’s stored isn’t some boring paperwork. In a real crisis, these details guide safety officers and firefighters to act fast and smart. All this matters more when you remember that chemicals cross borders and end up far from where they started.
Keep It Respectful, Keep It Safe
Handling Bismuth Neodecanoate safely keeps people healthy, companies out of regulators’ crosshairs, and the environment intact. It doesn’t take fancy systems or expensive technology—just discipline, the right gear, and respect for every bottle on the shelf.
Pushing Limits in Coatings and Paints
Coatings and paints benefit a lot from bismuth neodecanoate. This compound steps in as a safer alternative to lead-based driers, especially for those who want hard-drying and tough finishes. Customers want fewer health risks and tighter safety laws demand better options. Paint companies have shifted towards bismuth-based driers to avoid lead and cobalt. Quick-drying alkyd paints, varnishes for floors and furniture, and commercial primers see real-world results using this compound. Manufacturers choose it for lower toxicity, steady drying performance across temperatures, and compatibility with both oil- and solvent-based systems.
A Reliable Player in the Plastics Field
PVC manufacturers often use bismuth neodecanoate as a heat stabilizer during extrusion and molding. It protects the polymer against degradation when exposed to high temperatures. Plasticizers and stabilizers that keep consumer products safe from heavy metals appeal to companies, particularly those exporting to regions enforcing strict restrictions. Cable insulation and food packaging hit fewer regulatory roadblocks by switching to this compound, and production quality stays consistent batch after batch.
Medicine’s Quiet Ingredient
Bismuth neodecanoate pops up in pharmaceuticals, though it usually stays out of the spotlight. Certain drugs use bismuth compounds to target disorders affecting the stomach and intestines. Bismuth agents calm the lining in the digestive tract and curb the growth of some bacteria, such as H. pylori, which plays a role in ulcers. Its use rides on bismuth's reputation as a gentler choice compared to other metals once favored in medical chemistry. Creative chemists keep exploring its possibilities, especially as concerns about drug side effects grow.
Creating Clean and Safe Fuel
Fuel producers need catalysts that perform under tough conditions. Bismuth neodecanoate shows up as one of these helpers during the refining process, especially when making products like biodiesel. It helps break down raw feedstocks into useable fuel with fewer toxic leftovers. Renewable energy startups and established refineries eye it as they work to meet clean energy standards without unnecessary exposure to more hazardous metals. For people working in these facilities, there is less risk of running into heavy-metal health hazards.
Shaping Future-Focused Industry
My experience working with chemical suppliers and industrial clients points to tighter enforcement on hazardous substances in manufacturing. People often don’t realize just how carefully regulated chemicals have become. Bismuth neodecanoate steps up where safety and performance both matter. Whether it’s in a tin of gloss paint in your garage, the PVC pipes in your kitchen, the pill bottle in your medicine cabinet, or even diesel in a farm truck, this compound helps bridge the gap between robust results and responsible choices.
What stands out is that people and businesses no longer see health and environmental safety as a side issue. If you visit a factory that switched to bismuth-based additives, you’ll notice the difference in air quality and worker confidence. For companies, using safer alternatives isn’t just about checking off a regulatory box. It’s a straightforward answer to increasing scrutiny, and an easy step toward cleaner, future-friendly products. Real progress often happens in places few notice, and bismuth neodecanoate has found its niche as a quiet but important player in safer, smarter manufacturing.