Bismuth Hydroxide: A Down-to-Earth Look at a Lesser-Known Compound
Historical Development
Back in the early 1800s, chemists stumbled upon bismuth hydroxide while exploring the more recognizable faces of the periodic table. At that time, bismuth often trailed behind lead and tin—nations mined it, but they had less use for its quirky chemistry. Curiosity kept the science going, though, and by the later half of the 19th century, European researchers learned to work up bismuth solutions with a touch of alkali. Instead of tossing this milky precipitate aside, some clever minds chose to test it, sparking the first wave of interest in its potential for pharmacy and pigments. The old notebooks from those days lack the exact purity of today’s data sheets, but the curiosity they recorded fueled future experiments.
Product Overview
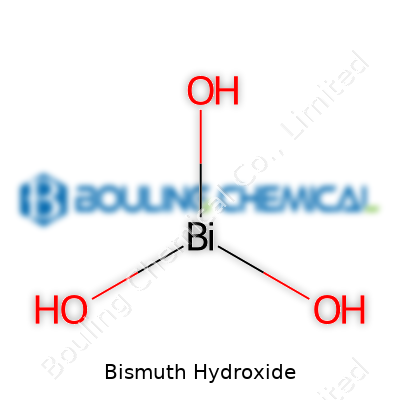
Chemists see bismuth hydroxide as a pale, almost white powder, the result of mixing a bismuth salt solution with an alkaline base. It carries the formula Bi(OH)3, though tricky sampling sometimes shows it with varying water content. In the modern market, bismuth hydroxide carves out a modest niche. It attracts attention as a safer alternative to more toxic heavy metal hydroxides. Its low solubility and mild base character open the door to specialty ceramics, cosmetics, and medical applications. You don’t see it stacked on the shelf next to table salt, but research labs and specialty producers keep a steady demand going.
Physical & Chemical Properties
On the lab bench, bismuth hydroxide shows itself as a fine powder, sometimes slightly yellow-tinted if contaminated with iron or other metal traces. It stays odorless. As you run your tests, you’ll see that it barely dissolves in water, isn’t flammable, and doesn’t corrode glass surfaces. Heating tends to drive off the water and generate bismuth oxide, a useful ceramic pigment in its own right. You mix it into acids, and it shifts into soluble bismuth salts. Strong bases may nudge it along into bismuthate complexes. The melting point isn’t a meaningful metric here, since heat decomposes bismuth hydroxide before a liquid phase emerges.
Technical Specifications & Labeling
Reputable producers back up bismuth hydroxide shipments with a certificate detailing purity (usually at least 98%), residual metals, pH range (typically mild alkaline in water), bulk density, and official batch numbers. Packaging calls for clear hazard labeling, noting it is not classed as hazardous under many transport rules but may still irritate eyes and mucous membranes. Scientific supply catalogs commonly list synonyms—“bismuth(III) hydroxide,” “tribismuth trihydroxide,” or simply “bismuth hydrate”—to avoid mix-ups.
Preparation Method
The classic method in labs dissolves a bismuth salt like bismuth nitrate in distilled water. Once you’ve got a clear solution, sodium hydroxide gets added dropwise. The hydroxide drops throw bismuth out of solution as a thick, white precipitate, which you can filter, rinse, and dry to collect your sample. Some industrial practices have switched over to continuous-flow reactors and automated pH control, chasing higher throughput and reproducibility. These setups scale up the wet method with better precision and cleaner yields. The reaction is familiar:
Bi(NO3)3 + 3NaOH → Bi(OH)3↓ + 3NaNO3.
Other bismuth salts serve as starting points, but the underlying chemistry doesn’t veer far from this reliable, waste-light reaction.
Chemical Reactions & Modifications
Chemists have leveraged bismuth hydroxide’s ease of conversion for decades. Heating it produces bismuth oxide, an important material in high-temperature superconductors and advanced ceramics. In acid, Bi(OH)3 dissolves to give bismuth salts like BiCl3 for use in specialty catalysts. Exposing it to strong oxidizers sometimes forms bismuth(V) compounds, rare in stable form but interesting for electronic materials research. Certain modifications, like treating Bi(OH)3 with organic ligands, create pharmaceutical intermediates that show bactericidal properties. Research teams continue to tweak these reactions to push the boundaries of what this low-key compound can do.
Synonyms & Product Names
Anyone hunting for bismuth hydroxide may find it sold under a handful of names. “Bismuth(III) hydroxide” is the strict IUPAC callout, though chemical catalogs just as often call it “bismuth hydrate” or “tribismuth trihydroxide.” Certain pharmaceutical grades go by trade names tied to proprietary blends, especially when paired with excipients for tablets or powders. Each alias traces back to the same core compound, so careful reading of the datasheet matters when purity, particle size, or intended use are on the line.
Safety & Operational Standards
Bismuth hydroxide doesn’t have the toxic bite of lead or mercury compounds, and that’s a big plus if you spend your days in the lab. Still, labs stick to responsible handling, with gloves and dust masks standard during weighing and mixing. Inhalation of fine powders, regardless of toxicity, irritates lungs and nasal passages. Water runoff from processes shouldn’t get dumped into sinks, since local bismuth concentrations can disrupt aquatic biology in high doses. Storage demands dry, sealed containers—moisture over time can start slow decomposition, especially if temperature swings in a storeroom.
Application Area
Medicine has long favored bismuth compounds for digestive health, and bismuth hydroxide contributes to this tradition in some stomach remedies and wound care agents. It gets work in pigment production, lending an opaque white to specialty paints without the hazards of lead. In technical fields, ceramicists value it as a precursor to advanced Bi-based oxides that feature in superconductors and oxygen sensors. Some cosmetic manufacturers tap it for hypoallergenic powders, since it gives a gentle finish while staying free from most known allergens. Electrochemists and material scientists see it as a jumping-off point for sensor and filter development. Although the market isn’t huge, the niches it fills remain important and hard to replace.
Research & Development
R&D teams dig deep into bismuth hydroxide because it sits at crossroads between moderate safety and surprising reactivity. In my own lab work, we explored its potential for targeted drug delivery coatings, drawn in by its straightforward chemistry. Researchers worldwide test how it might capture toxic anions or serve as scaffolding in new nano-structured catalysts. New studies into bismuth’s interaction with biological systems keep bringing fresh ideas for both diagnostics and therapies. Industrial labs focus on cleaner production, trying to minimize metals waste and dial in tighter particle size controls. Collaboration between university groups and specialty firms keeps the pace of discovery steady—even if the headlines stay rare outside chemistry circles.
Toxicity Research
Bismuth hydroxide is less notorious than lead or arsenic, but thorough toxicological testing still matters. Studies in mammals show limited absorption through the gastrointestinal tract and an ability to clear smaller doses without major organ damage. Long-term exposure studies look for subtle neurological or renal impacts, especially with repeated ingestion or inhalation, and so far the data suggests a wide margin of safety for industrial or medical use at approved concentrations. Regulatory agencies keep pressure on manufacturers to update risk assessments as research continues, especially as new applications stretch the limits of exposure. Environmental studies track bismuth runoff from factories, noting that while acute toxicity stays low in most species, the true ecosystem impacts of long-term accumulation deserve more investigation.
Future Prospects
Bismuth hydroxide stands ready for more action, as green chemistry principles push industries away from more toxic alternatives. Scientists explore its use in antimicrobial surfaces, eco-friendly pigments, and as an intermediate in the search for new superconducting materials. Medical research shows a slow but steady uptick in patents for controlled-release pharmaceuticals using bismuth hydroxide matrices. Its gentle reactivity, decent stability, and relative environmental friendliness could secure it a place in tomorrow’s materials toolbox. Creative minds in the lab might soon find entirely new uses for this old but underappreciated compound—if so, history suggests demand could surge from an unexpected direction, just as it did the first time chemists gave it a second look.
Inside the Lab and Out in the World
Bismuth hydroxide often sits forgotten on chemistry shelves unless you know what to look for. In medical circles, this humble compound helps doctors calm upset stomachs. Think of old-school remedies—bismuth hydroxide stands behind many of those chalky antacid medicines lining drugstore shelves. I remember my grandmother trusting her pink remedy, never asking about the chemistry behind it. Turns out, bismuth hydroxide plays a key role in relieving stomach irritation and even treating mild digestive disorders.
Medicinal Uses You’ll Actually See
The most common way people encounter bismuth hydroxide is through its role in bismuth subsalicylate, the ingredient powering popular stomach-settling liquids and tablets. This molecule forms when bismuth hydroxide reacts with salicylic acid. Scientists started using this combo because it coats the stomach lining, eases discomfort, and fights off some nasty bacteria in the gut. Research from the National Institutes of Health even points to bismuth-based compounds helping treat infections caused by Helicobacter pylori, a big driver of peptic ulcers.
Beyond Medicine: What Else Gets Help from Bismuth Hydroxide?
Pharmaceuticals only show part of the story. I first noticed bismuth hydroxide outside the medicine cabinet during a stint in a college ceramics workshop. Potters and ceramicists value bismuth for the color and finish it brings to glazes. Bismuth hydroxide acts as a precursor for a range of bismuth salts and oxides. Because it resists corrosion and handles high heat, you’ll spot it in specialized pigments and laboratory glassware.
Chemical industries use bismuth hydroxide to help pull heavy metals out of wastewater. In settings where traces of lead or other toxins need cleaning, engineers use bismuth hydroxide as a filter—kind of like a specialized sponge. Since bismuth is considered much safer compared to lead and mercury, it’s become popular where health and environmental concerns come first.
Potential Risks and How We Handle Them
Not every bismuth-based product walks straight to the shelf. Safety agencies keep watch on all compounds added to medicines and industrial processes. A headline in recent years focused on rare side effects: if someone consumes too much bismuth over time, the metal can build up and cause complications affecting the kidneys or nervous system. Keeping tabs on dosages and proper disposal, especially in medical and industrial uses, keeps these risks in check.
One way to limit these problems involves stronger monitoring in factories and medical settings. Strict records, good training, and improved wastewater filters can help companies use bismuth wisely. Clinicians track symptoms in patients who take bismuth-based drugs, so long-term issues get caught early. Over-the-counter regulations already set safe limits for consumers.
Looking Ahead: Greener Chemistry and Smarter Use
Interest in green chemistry continues to push for safer, less wasteful materials. Bismuth hydroxide already offers a less toxic alternative compared to older, harsher chemicals, but scientists keep searching for ways to improve its production and cut environmental impact. From medicine chests to water treatment, bismuth hydroxide’s reputation as a reliable, relatively safe agent has kept it relevant for over a century. It's worth keeping an eye on as research teams around the world unlock new uses for this old compound.
What’s In a Name?
Bismuth Hydroxide doesn’t show up much in everyday conversation, but you’ll spot it in labs and a few specialty industries. The chemical formula stands out as Bi(OH)3. This tells us each molecule ties together one bismuth atom and three hydroxide groups. Bismuth sits below antimony on the periodic table, slotted among metals, but unlike lead or mercury, it’s far less toxic. Even so, it doesn’t get the attention of copper or zinc.
Why Bismuth Hydroxide Matters
Bismuth Hydroxide isn’t found in the medicine cabinet in pure form, but parts of the pharmacy trace back to it. Take bismuth subsalicylate, the pink liquid for upset stomachs—bismuth hydroxide plays a key role early in its creation. Inorganic chemists count on Bi(OH)3 for producing other bismuth-based compounds or for lab demonstrations about solubility and precipitation.
Getting Down to Chemistry
This compound pops up most often as a white, bulky precipitate. If you add a strong base like sodium hydroxide to a solution with bismuth nitrate, you’ll see a cloudy solid—this is bismuth hydroxide forming on the spot. It’s technically sparingly soluble in water, meaning it doesn’t disappear right away like table salt. If you keep increasing the base, some of the hydroxide dissolves again to create more complex ions, a detail prized in analytical chemistry.
From a safety perspective, Bi(OH)3 is much gentler than many heavy metal hydroxides. The Environmental Protection Agency doesn’t list it among substances with major health risks, at least not in the amounts any ordinary consumer would encounter. That doesn’t mean it’s edible, but it lowers the stress for people in labs.
Bismuth’s Green Reputation
When environmental agencies started clamping down on poisonous heavy metals, industries hunted for safer alternatives. Bismuth, including in forms like hydroxides, took up the slack where lead compounds once reigned. Fireworks, cosmetics, and pigments all benefit from this shift. Even today, tech companies eye bismuth hydroxides and oxides while researching new lead-free solders.
Some paints and coatings turn to bismuth hydroxide for non-toxic anti-bacterial properties. This trend links back to public health. We’ve seen enough stories about toxic lead in paint chips or mercury in wastewater to know the need for safer options isn’t going away.
Challenges and Ways Forward
Bismuth itself doesn’t come cheap, which limits the spread of bismuth hydroxide in mass production. Mines that produce bismuth often do so as a byproduct of copper, tin, or lead, meaning supply fluctuates with broader economic trends. Mining also pulls local communities into tough conversations about labor and land use. In my time around mining towns, you hear real concerns about keeping water clean or making sure jobs stick around as tech changes.
Recycling can help. Europe and Japan lead in pulling bismuth out of scrap electronics. Pushing for better recycling laws, and encouraging manufacturers to use traceable, recycled metals, fits the growing demand for ethical sourcing. Universities, too, have a role: research on synthesizing bismuth hydroxide directly from mining slurry or e-waste could shift the game.
With more companies listing life-cycle data for their chemicals, buyers can pressure suppliers to choose safer and more sustainable ingredients. Bismuth Hydroxide’s low toxicity and potential for recycling put it on the radar for any company hoping to balance productivity with environmental care.
Understanding What You’re Dealing With
Bismuth hydroxide pops up in classrooms and laboratories where folks work with basic chemistry. The material is a white, chalky powder that can seem pretty tame on the surface. People sometimes treat it as a safer alternative to other heavy metal compounds, because bismuth doesn’t have the terrible reputation of lead or mercury. That reputation comes from real evidence; most of the research calls bismuth compounds among the more "gentle" choices in the heavy metal world. Oddly enough, bismuth even shows up in stomach remedies you can find at the local pharmacy.
Low Toxicity—But Not a Free Pass
In my own work, bismuth hydroxide made its way into bench-side testing a few times. Nobody ever wore hazmat gear to handle it, but everyone wore gloves. The reason is pretty simple: even though the science says bismuth is low in toxicity, it’s still a heavy metal compound. That alone signals, keep your exposure as low as possible. Peer-reviewed studies back up the claim that bismuth’s toxicity sits far below its cousins—lead and mercury can wreak havoc on your nerves and organs, while bismuth tends to spare people from lasting harm.
Looking at actual safety documents, bismuth hydroxide can irritate skin, eyes, or the lungs if you inhale the dust. That matches my own experience; if the container gets bumped and the powder forms a little cloud, people cough and start waving away the air. The Material Safety Data Sheet recommends standard precautions. You want gloves, goggles, and a dust mask when you scoop or weigh the powder. Anything less feels careless, because all those lab powders carry some risk you don’t see or smell.
Don’t Treat Lab Hygiene Casually
Many students and some eager interns figure a low-hazard powder like bismuth hydroxide means you can skip cleanup. That’s an easy way to turn a safe chemical into a mess. Spilled powder in a drawer, or on your hands, increases the chance that you swallow a little by accident. Nobody gets sick right away, but repeated exposure is hard to track over months. I’ve seen old stains on the workbench where nobody can say for sure what’s in them. That’s not a good practice for a shared lab or a classroom where mistakes spread quickly.
Science Backs Careful Practice
Bismuth as an element tends to stay in the gut and leave the body quickly, which helps explain why major poisonings rarely show up in medical journals. Still, medical case reports exist where chronic bismuth use led to nerve issues or kidney trouble, though these happened with unusual amounts and lasted exposures. The risk goes up when powders go airborne or get washed down the sink, heading into the drainpipes. Even harmless-sounding compounds add up in waterways, raising questions for the environment.
Practical Steps for Peace of Mind
Simple habits take care of most concerns. Store bismuth hydroxide in a sealed container. Always use gloves, eye protection, and a dust mask if you might stir up powder. Wet-wipe your workspace afterward instead of brushing powder around. Never eat or drink nearby, and keep a routine for washing hands before you leave. If you ever spill the powder, skip sweeping and pick it up with damp towels or disposable wipes, then toss them in the chemical waste bin.
The biggest lesson: calling a chemical “safe” doesn’t mean “thoughtless.” Strong habits learned with less-toxic materials carry over if you ever need to handle nastier chemicals, and protecting yourself isn’t just a nice idea—it's a real skill, backed by years of science and plenty of common sense.
Getting Right Into the Storage Basics
Bismuth hydroxide plays a role in labs, sometimes clinics, and even pops up in some chemist’s stocks, especially where safe, low-toxicity compounds are important. Still, keeping it safe doesn’t just protect inventory. It also prevents accidents. I’ve watched enough hurried technicians skip steps, only to spend the next week cleaning up a spill or handling corroded containers. Simple vigilance prevents most mishaps.
Choosing the Right Container
Direct exposure to air or light messes with bismuth hydroxide’s chemical structure. Glass with a solid seal stands as the container of choice. Plastic stores don’t always cut it because strong bases interact with certain plastics and caps loosen over time. If you’ve ever opened a loosely shut bottle, you know the mess that follows. Glass bottles with ground-glass stoppers work best for long-term stability.
Shielding from Moisture and Contaminants
Bismuth hydroxide reacts with carbon dioxide and wears down in humid environments. Storing it in a dry cupboard, away from running sinks and chemical washes, minimizes this. Those little silica gel packets you sometimes find in shipping boxes? Tucking one next to the bottle in a sealed cabinet handles unwanted moisture. Avoid keeping it near any acids, since cross-contamination ruins both compounds and damages workspaces.
Protecting from Temperature Changes
Sharp temperature swings speed up decomposition and cause container cracks—no one wants to clean a sticky shelf. Room temperature, usually between 20°C to 25°C, keeps bismuth hydroxide stable. Avoid storing bottles above stoves or in sunlit windows. My old supervisor lost two jars that way, both oozed crystals by month’s end.
Clear Labeling and Inventory
Mislabeling causes confusion and can spark chain reactions of errors. Every container deserves a clear, waterproof label: full name, date acquired, and any hazard info. Tracking this in an inventory sheet helps avoid doubling up or using old, degraded stock. One slip-up with expired material can throw off a whole project’s results. If a label begins to fade, fix it right away instead of waiting.
Addressing Spills and Disposal
If a spill happens, gloves and lab coats must go on. Use a brush and dustpan, no vacuum cleaners, then wipe the surface down with a damp disposable cloth. I always keep a dedicated waste bottle, labeled 'bismuth waste,' so nothing gets mixed or poured down the drain. For larger quantities, a hazardous waste company takes over—no need for shortcuts. The EPA and local agencies have rules for metal-bearing waste, and following them keeps your workplace out of trouble.
Training and Supervision Set the Standard
Not every new lab tech has hands-on chemical storage experience. Senior staff members—really, anyone with more than a few months in the lab—need to walk rookies through storage procedures. Running mock drills and quick reviews builds confidence. Ignoring routine checks leads to lazy shortcuts, which eventually cause problems harder to fix than storing the chemical properly the first time.
Looking Forward
Safe bismuth hydroxide storage isn’t only about tidy shelves. It’s about responsibility—to colleagues, research, and yourself. Proper habits form the backbone of every solid lab. If one person slacks off, everyone eventually feels the effects. Keeping these strategies in place preserves both safety and the quality of your results, every single day.
Getting to Know Bismuth Hydroxide
Bismuth hydroxide often lands on the chemist’s bench because it looks just as interesting as it acts. In the lab, it shows up as a heavy white solid, powdery to the touch. As a person who has wrestled test tubes and weighed many crystalline powders, I can say bismuth hydroxide doesn’t fade into the background. Even after you’ve finished working with it, bismuth hydroxide leaves stubborn traces; it tends to cling to beakers and spatulas, absolutely refusing a quick rinse with water.
Color, Texture, and Feel
With a soft white appearance, fresh bismuth hydroxide feels dense but not gritty. It doesn’t pack loosely like chalk dust or all-purpose flour—it settles as a fine powder. Many textbooks mention how it sometimes turns yellow due to slight oxidation, and that's accurate. If left exposed or sitting in humid air, a yellowish tint creeps in, hinting that bismuth doesn’t like being ignored.
Solubility and Stability
In real-world work, bismuth hydroxide just doesn’t want to dissolve in plain water. Even with plenty of stirring, water and this powder won’t mix. This property actually makes it easier to handle in the lab. If you spill some, a quick wipe is enough, and you don’t end up chasing invisible drips across the bench. Solubility does change, though, with acids and bases. Drop some hydrochloric acid onto this powder, and it fizzles away—quickly turning into bismuth chloride, usually with some heat involved. Add extra base, and you get complex bismuthate ions instead. For anyone doing practical chemistry, these reactions aren’t just facts—they keep your day moving.
Melting, Decomposition, and Usage Limits
Try to heat bismuth hydroxide, and it will let you down quickly. There isn’t a well-defined melting point. Bismuth hydroxide prefers to break down before it gives you a liquid puddle. Heat drives off water and leaves bismuth oxide—a yellow powder that stains everything it touches. I’ve learned not to overdo things with the Bunsen burner here unless you want to clean up a mess that’s trickier than before.
Crystal Structure and What It Means
Unlike some flashier materials, bismuth hydroxide rarely shows off sparkling crystals with ease. Typically, you get an amorphous or poorly crystalline solid. For people concerned with purity or uniform size, this can be frustrating. On an atomic scale, there’s a slightly disordered arrangement—enough that researchers still puzzle out the details decades after it was first made. For applications in medicine or electronics, this limits how it can be used. It also explains why you don’t see it flooding the commercial market the way some materials do.
Where the Limits Lead—Potential Solutions
Controlling humidity can help slow down the yellowing process. Lab techs put containers of bismuth hydroxide in a desiccator, using silica gel or other moisture traps. For the stubborn residue problem, a diluted acid wash (under safe conditions and with protective gear) clears it out without damaging glassware. Anyone hoping to use it in new tech should focus on stabilizing its chemical form and finding better methods to control its crystal structure. Looking back, every improvement in lab handling—better containers, smarter use of pH, improved purification—has come from facing these stubborn facts head-on instead of hoping they vanish.