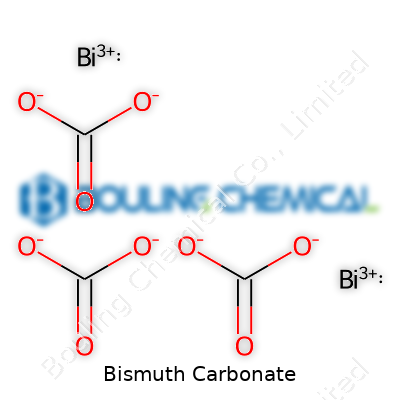
Bismuth Carbonate: A Down-to-Earth Examination
Historical Development
Centuries ago, alchemists kept only rough records of what later generations would name bismuth carbonate. Much of what people now know about this compound comes from the late 1700s and early 1800s, as scientists in Europe dug into the chemistry of strange white powders. In those early days, confusion with lead and tin was common. Names shifted as researchers figured out exactly what they had made, before modern nomenclature settled things. This humble compound quietly worked its way from dusty laboratory discoveries toward actual industrial and medical application. Today, bismuth carbonate stands far less mysterious, though its role in industry remains important for several sectors.
Product Overview
Bismuth carbonate often appears as a fine, white or pale yellow powder, with a slightly pearlescent sheen. Many use it as a source of bismuth in ceramic glazes, pigments, and medicines. Its physical appearance can mask its weight. People find it surprisingly heavy for a powder, which matches the element’s position low on the periodic table. The medical field recognized its stomach-soothing effects long ago, building on folk remedies. In chemical supply catalogs, you’ll find a range of grades, each intended for specific end uses, such as pharmaceuticals, lab reagents, fine ceramics, or catalysts.
Physical & Chemical Properties
Bismuth carbonate, with the formula Bi2(CO3)3, holds onto water molecules in its crystal structure and forms a slightly basic compound, breaking down in acids to release carbon dioxide. The material avoids solubility in pure water, so it persists in suspension rather than dissolving away—a fact that informs both its usage and handling. Chemically, bismuth sits in a unique spot between heavy and benign elements, making this compound less reactive and less toxic than many lead or cadmium salts. You’ll find that bismuth carbonate’s density packs more heft per scoop than most comparable white powders, and it transitions through decomposition to bismuth oxide upon heating, giving it a role in ceramic production.
Technical Specifications & Labeling
Labs, pharmacies, and manufacturers label bismuth carbonate with purity, hydrate status, mesh size, and trace impurities like lead and arsenic. Pharmaceutical grades must meet pharmacopeial limits for heavy metals, while industrial material calls for batch consistency and predictable particle size. Labels show warnings for dust inhalation and safe storage instructions—dry, airtight containers, away from acids that could break down the powder. The packaging often includes CAS, EC numbers, and supplier batch codes to trace a container’s origin. Rules require keeping the compound’s documentation up to date, especially when the use crosses into food or medical territory.
Preparation Method
Chemists prepare bismuth carbonate by reacting a soluble bismuth salt—often bismuth nitrate—with a carbonate source. Sodium or potassium carbonate solutions introduce carbonate ions, immediately causing the bismuth to drop out as a heavy, cloudy precipitate. Filtration, washing, and drying steps refine the crude product. Some processes control temperature or the amount of reactants for finer grain or better purity. At a small scale, chemists rely on glassware and good agitation to avoid clumps, and scale-up for manufacturing needs open tanks, industrial mixers, and massive filtration rigs. The byproducts—commonly soluble alkali nitrates—wash away with repeated rinsing.
Chemical Reactions & Modifications
Bismuth carbonate enters other chemical transformations with gentle prodding. Strong acids decompose it, bubbling carbon dioxide and leaving soluble bismuth salts behind. Controlled heating pushes it to bismuth oxide, allowing ceramists to produce glazes or electronic materials. In some synthesis, bismuth carbonate serves as a starting point for organobismuth compounds, which find roles as laboratory reagents or specialty materials. Modification with surfactants or dispersants can change particle behavior for coatings or composite manufacture, expanding its application set beyond its old roles as pigment base or antacid ingredient.
Synonyms & Product Names
This material answers to several names in commerce and research. Among them: bismuth subcarbonate, basic bismuth carbonate, and by various hydrated forms such as bismuth oxycarbonate. Catalogs may include obsolete or trade names, especially if the product aims at the pharmaceutical or ceramic market. International suppliers list it in translated forms, depending on local chemical naming conventions. For regulatory and shipping purposes, batch codes and abbreviations appear alongside the full English or Latin names, connecting shipments back to batch records and certificates of analysis.
Safety & Operational Standards
People handling bismuth carbonate need to understand the basic precautions: keep dust down, avoid breathing the powder, wash skin and work areas. Gloves and dust masks (or better, a fume hood) should be a matter of habit even though this compound lacks the menace of heavier metals. Pharmaceutical producers put extra steps in place—filtered air, closed transfer systems, and rigorous quality control checks. Hazard communication emphasizes both immediate risks (eye and nose irritation) and long-term ones (possible but rare heavy metal accumulation in poorly managed operations). Garbage disposal follows chemical waste routes, never regular trash, honoring environmental rules on bismuth.
Application Area
You’ll find bismuth carbonate wherever gentle white pigments matter. Ceramics use the powder to form glossy white glazes, taking advantage of the compound’s ability to break down and “flux” at moderate kiln temperatures. Over-the-counter stomach medicines rely on bismuth carbonate’s soothing and anti-microbial properties—a function documented for decades in clinical practice. Rubber and plastics manufacturers reach for it as a stabilizer or pigment, benefiting from its lack of color shift under heat and sunlight. Some researchers use it as a precursor to more exotic bismuth compounds, exploiting its mild reactivity and relative safety.
Research & Development
Active research on bismuth carbonate targets eco-friendly materials. Chemical engineers want to push into lead-free ceramics and solder, using bismuth as a safer heavy metal. Nanotechnologists look for new synthesis methods producing particles at the smallest scales, enabling applications in electronics, catalysis, and medicine. Drug development sees new interest in bismuth salt formulations and testing, propelled by antibiotic resistance issues and the need for alternative treatments for stomach infections. Efforts to improve manufacturing processes focus on clean, low-waste reactions and developing ways to keep purity up while costs and energy use drop.
Toxicity Research
Compared to most metal carbonates, bismuth carbonate presents a lower health risk profile. Research indicates limited absorption through the gut, and the human body sheds bismuth more readily than lead or cadmium. Still, animal studies urge caution, as very high, sustained doses over long periods do lead to mild kidney disruption and tissue accumulation. Most clinical settings see few side effects—occasional digestive complaints, rarely hypersensitivity reactions. Despite these reassurances, keeping exposure minimal matches modern safety standards. This means avoidance of food contamination, limited airborne dust, and medical supervision during therapeutic use.
Future Prospects
Bismuth carbonate faces a mixed future. On one hand, increasing pressure to abandon toxic heavy metals in industrial and electronic materials favors bismuth compounds as safer alternatives. Medical research keeps the compound relevant as bacterial resistance and gastrointestinal disease rates push doctors to revisit bismuth-based treatments. Challenges stem from supply limits and competition with cheaper, more familiar fillers and pigments. The compound’s sustainability story depends on improved mining, processing, and efficient downstream use. Those advances, supported by regulatory checks and responsive industry standards, shape whether bismuth carbonate moves from niche to mainstream in the next wave of green chemistry and advanced manufacturing.
Medicine and Traditional Uses
Bismuth carbonate often sits in the back row in the world of compounds, but doctors and pharmacists haven’t forgotten its place on the shelf. It pops up in treatments for stomach troubles. People have used medicines with bismuth salts for generations when heartburn, diarrhea, or ulcers strike. If you’ve seen old patent medicine bottles, there’s a good chance bismuth was an ingredient. Now, you find its cousins like bismuth subsalicylate in well-known over-the-counter products. Chemically, bismuth carbonate changes in the stomach to help coat and protect that irritated lining. For those who struggle with sensitive digestion or long-term stomach issues, sometimes this kind of gentle mineral compound makes all the difference.
Industry and Specialty Applications
In industry, bismuth carbonate rarely stands alone. Ceramics look brighter and glossier because of it. Glassmakers blend it in for special finishes or to cancel out unwanted colors. Manufacturers searching for a lead-free pigment option appreciate its low toxicity compared to other heavy metals. Bismuth compounds manage to squeeze into fireworks, making colored flames without the hazards of older chemicals. As green chemistry gets more attention, safer alternatives for pigments and coatings matter more every year. Personally, after handling many types of glazes in an art studio, those labeled “bismuth” always seemed less irritating than older formulas with lead or cadmium. Artists appreciate the lower risk, especially in school settings where safety comes first.
Laboratory Use and Research
Chemists keep bismuth carbonate handy for its reactivity. It helps produce other bismuth-based chemicals – useful in everything from semiconductors to catalysts. In research labs, it’s a starter for more complex synthesis projects. Bismuth itself resists corrosion and has a low toxicity profile. That combination means new applications pop up as scientists search for better alternatives to toxic elements. For anyone working with kids or pets in the home lab, having safer chemicals around is a relief. Picking bismuth carbonate over some of its riskier peers just makes sense for safety and environmental impact.
Safety and Environmental Concerns
Bismuth’s reputation as a safer heavy metal stands on solid evidence. Studies have shown its low absorption by the body and limited ecological harm compared to metals like lead or mercury. Several major health organizations list bismuth salts as safe in small doses, which gave pharmacists confidence back in the days before strict regulations. Still, nothing is completely harmless—misuse or inhalation of the dust can cause health problems. Industry workers follow guidelines to avoid breathing in powders, and any spills get cleaned with care. Growing up in a mining region, local stories about old-timers exposed to all sorts of chemicals drove home the value of safer materials. Progress depends on safety as much as science.
Outlook and Better Solutions
Bismuth carbonate might never headline mainstream news, but its role stretches far beyond the laboratory. Pushes for lead-free products and safer pigments continue to grow. More research into how bismuth fits into medical, art, and tech fields can unlock solutions that protect both people and ecosystems. Prioritizing education, proper storage, and smart product design helps keep bismuth carbonate working quietly in the background, supporting daily life and future innovation.
What Actually Is Bismuth Carbonate?
Bismuth carbonate lands in chemistry books as a white, powdery compound made from bismuth and carbonate. It shows up in some medicines and even in certain cosmetics. Walk into a pharmacy and many folks know bismuth as the stuff behind pink stomach medicines—though those products use bismuth subsalicylate, not bismuth carbonate. The curiosity kicks in with this: why would bismuth carbonate end up in something people put in or on their bodies?
Safety Record and Use in Medicine
Bismuth in different forms isn’t new in folk and mainstream medicine. Doctors have prescribed bismuth compounds for upset stomachs, some infections, and even as a treatment for a few tropical bugs. Laboratories test compounds like bismuth carbonate to check how they react with the body and other chemicals.
Direct evidence about bismuth carbonate’s safety remains thin. Most studies focus on bismuth subsalicylate or other salts. There’s no green light from food or drug regulators for swallowing bismuth carbonate in the same way that antacids or painkillers get approved. That matters, because the structure of a chemical can change how the body deals with it.
What Happens in the Body?
Bismuth salts can pack a punch if you eat or inhale too much. People exposed to heavy bismuth levels have turned up at clinics with confusion, kidney trouble, or dark lines on their gums. The body doesn’t use bismuth for anything essential. Most people flush out small amounts with urine and stool, but bigger doses stick around longer and may cause harm.
Based on research and my own reading in biochemistry, the body reacts differently to every bismuth compound. Bismuth carbonate isn’t as well studied as its cousin in pink stomach medicine. That absence of clear evidence sets off warning bells in anyone who values their health. Unproven claims from supplement sellers or sketchy online vendors should signal caution. Valid safety claims need to rest on clinical testing, not promises.
Common-Sense Takeaways
If a compound only shows up on ingredient labels outside pharmacies, people should ask more questions. Certified medicines with bismuth salts earn their badges after long clinical trials that prove safety and effectiveness. Pharmaceutical companies and government agencies keep tabs on dosing, side effects, and long-term health impacts. No such checkpoints stand for bismuth carbonate sold as a raw compound or online supplement.
Some bismuth compounds do soothe stomach pain or treat mild infections, but bismuth carbonate isn’t a household name among doctors. Noble claims of detox or wellness benefits swirl online, yet none of those stand up in peer-reviewed journals. The lack of quality research on bismuth carbonate for internal use shows the risks don’t match the supposed rewards.
Better Paths Forward
Regulators, researchers, and consumers play a part in keeping things safe. Independent labs need to test bismuth carbonate for safety if anyone hopes to use it internally. Transparent labeling and honest marketing should steer people away from risky products. If stomach or skin trouble hits, it’s safer to stick to time-tested medicines or ask a qualified doctor for advice. Reading between the lines on product labels and trusting peer-reviewed data always beats rolling the dice with a chemical.
The Reality of Bismuth Carbonate in Everyday Labs
Bismuth carbonate feels hard to ignore for anyone poking around in chemistry. It looks like a simple white powder, but its character tells a bigger story. I’ve watched students in the lab question where the odd fizzing comes from. Drop it into an acid, and you spot bubbles. That’s not just for show. Bismuth carbonate, with its formula Bi2(CO3)3, reacts instantly with acids, breaking down to release carbon dioxide. No need for fancy explanations — just a clear sign this stuff doesn’t like strong acids hanging around. Carbonate ions in the compound give up their gas, leaving bismuth salts behind. This reaction marks out bismuth carbonate from a crowd of duller compounds.
Stability and Temperature Sensitivity
Chemically, this powder keeps its cool under a dry atmosphere, away from strong light and moisture. In my own experience, try leaving some in a humid corner and you’ll spot clumping and slow dulling. Bismuth carbonate doesn’t dissolve easily in water. High temperatures break it down, driving off carbon dioxide and producing bismuth oxide. That’s not just trivia — thermal decomposition forms the basis for several industrial syntheses. Warm it up enough, and you’ll watch a clear color change, with the white powder fading into yellowish hues.
Other Chemicals: Friends and Foes
Bismuth carbonate shrugs off mild bases. Expose it to alkalis, and you can sometimes coax it into solution, but the process drags, rarely neat or tidy. Strong acids remain the surest way to bust it apart. You see similar reactions with other carbonates, but with bismuth you get unique byproducts. This gives labs an efficient route to form bismuth-based drugs or catalysts without the sideline waste common with less well-behaved metals.
Uses Connected to Properties
You can trace its use as a mild antacid and a source for other bismuth compounds straight back to the way it reacts with acids and heat. In the pharmacy world, that consistent fizz helps buffer stomach acid and settle nerves, especially when mixed with other soothing ingredients. Not many inorganic compounds tread into medicinal use, so bismuth carbonate stands out for its balance — not too reactive in the gut, but responsive enough to switch up when needed.
Environmental and Health Considerations
Bismuth itself wears the badge of "least toxic heavy metal," so worries don’t run high compared to lead or mercury. But chemistry teaches you to stay careful. Powders mean dust, and dust means it ends up in your lungs if you get sloppy. In labs and factories, simple face masks and good ventilation knock down this risk. Regular disposal guidelines in the EU and US skew cautious, not because bismuth carbonate is a top-tier hazard, but because consistency protects everyone down the line.
What Could Make Bismuth Carbonate Better?
Purity tends to slip in large batches. Tiny traces of lead from ores sometimes sneak in — that’s a genuine challenge. Better refining and tighter sourcing standards form the only real shield. Over the years, I’ve seen great improvements with supplier transparency. One simple label, tracing the batch back to its ore, saves endless paperwork and worry for both suppliers and end users. Shelf life extends if kept dry and away from sunlight, so clear storage instructions help keep the compound working as intended.
Why Safe Storage Matters
Managing chemicals asks for more than ticking boxes; it demands clear care and a bit of know-how. Bismuth carbonate gets used in labs, ceramics, even medicine manufacturing. I remember working in a university research lab where shelving and labeling made all the difference between predictable workdays and last-minute accidents. Secure storage meant students could focus on their projects, not worry about handling hazards.
Understanding Bismuth Carbonate’s Properties
Bismuth carbonate usually shows up as a white or yellowish powder. It doesn’t dissolve much in water. It doesn’t ignite easily, but like most powders, it tends to drift in air if spilled. Some folks forget that even non-flammable powders can bring health risks if inhaled or mixed up with acids. The powder may give off carbon dioxide in an acid spill, so some basic awareness lowers risk in busy workspaces.
Right Storage Conditions
Good habits start with temperature. Keep the container in a cool, dry spot. Moisture leads to clumping and might trigger slow chemical changes, which turn a handy material into a mess or a safety headache. Humid storage rooms turn powders into caked lumps. In high school, our chemistry storeroom sat right next to old, leaky pipes. Keeping silica gel packets around the jars avoided nasty surprises.
Store the carbonate away from sunlight and strong lighting. UV breaks down many substances, and although bismuth carbonate holds up better than some, there’s no reason to test its limits. Even strong overhead lights can speed up reactions, so shelves behind closed cabinet doors work best. Make sure the area has enough airflow. That keeps any stray dust from building up and doubles as preparation in case of a minor spill.
Choosing the Right Containers
Glass jars with tight-fitting lids give the best protection. The old amber glass type blocks stray light, keeps out sneaky humidity, and won’t react with the powder. Polyethylene bottles work too, but avoid storing the powder in metal. Even with bismuth’s low reactivity, common sense says never to mix chemicals and metals unless you know the full reaction story.
Label every jar with clear, big writing. No torn tape, faded marker, or sticky notes. Everyone in a shared space deserves to know what they're handling, and proper labeling saves time during periodic inspections or clean-outs.
Take Safety Seriously
Bismuth carbonate doesn’t strike fear into the hearts of chemists the way some acids do. Still, fine powders cause breathing trouble and irritation. Spilled chemicals can turn minor slip-ups into drawn-out cleanup sessions. Store the powder out of reach of kids, pets, and anyone without proper training.
Keep personal protective gear nearby—dust masks and safety glasses at the minimum. If possible, place the containers on shelves lower than head height. That single choice once stopped me from getting a powdery blast to the face during a minor earthquake.
Emergency plans don’t get used often, but they matter. Know the nearest eyewash station, and post directions on handling accidental spills—wipe up with a damp cloth, not a broom. Skip water if acid contamination is possible and use neutralizers suited to the chemicals stored nearby.
Improving Storage Practices
Rotation keeps supplies fresh. Use older stock before opening new containers. Check expiration dates, even for simple compounds like bismuth carbonate. Small habits, such as having a logbook near the storage area, improve traceability.
Train every lab member—including newcomers and occasional helpers. Brief everyone on storage protocols during orientation. Preventing trouble costs much less than fixing it, and the local fire inspector always sleeps better when clear standards get followed.
Bismuth carbonate stays simple to handle, as long as you treat it with steady respect. Store it right, and it remains just another tool at your disposal.
Understanding Why Dosage Matters
Bismuth carbonate has carved out a niche in specialized areas like pharmaceuticals and industrial chemistry. Any substance with bioactive properties needs careful application. Bismuth salts, including the carbonate form, have a rich history in medicine—especially for dealing with digestion issues and infection control. Knowing the dosage and right way to apply it keeps users safe and supports positive outcomes.
What Doctors Say About Safe Use
Medical guidance trumps any general advice. Doctors and pharmacists look at a patient’s age, health history, and the intended use before recommending a dose. In medical settings, bismuth carbonate hasn’t received the same household familiarity as bismuth subsalicylate (the active ingredient in some over-the-counter stomach remedies). Still, some compounds with bismuth carbonate find use in compounding treatments for infections such as Helicobacter pylori. Here, dosage isn’t picked off a chart online—the healthcare provider chooses based on clinical need, weight, and possible interactions with other medications.
Guiding Principles in Pharmaceutical Usage
Bismuth carbonate’s pharmaceutical role ties back to an understanding of how it works in the body. Generally, formulations aim for a daily bismuth dose between 120 mg and 500 mg, but this is a reference point—not a prescription. Higher concentrations risk side effects, ranging from discoloration of the tongue to more serious kidney or neurological complications. Long-term or excessive use sometimes leads to buildup in the body, and people with kidney trouble should avoid bismuth salts entirely. If someone faces persistent stomach symptoms or seeks “natural” alternatives, discussion with a healthcare provider avoids needless risks. Self-treating with unregulated doses doesn’t pay off and can mask deeper issues.
Industrial and Laboratory Settings
On the chemistry bench, technicians measure bismuth carbonate differently. Here, dosages aren’t about milligrams per body weight but concentration in a mixture or solution. Protective gear and fume hoods become more important than any calculation, as exposure through skin or inhalation brings its own risks. Factories and laboratories follow occupational safety rules, and each process has protocols based on toxicity assessments published in databases like the National Institute for Occupational Safety and Health (NIOSH).
Real Challenges With Overuse and Regulation
Sometimes manufacturers or online supplement outlets sidestep established pharmacy standards. Powdered compounds listed online might not include reliable dosage advice or risk statements. Few customers realize that products labeled as “bismuth carbonate” can come in varying grades—pharmaceutical, analytical, or technical. Using the wrong grade, or guessing at a dose, sometimes leads to more harm than help. Calls to poison centers related to bismuth join a long list of preventable incidents. Regulatory bodies work to address these risks, but enforcement still lags in some regions. Public education campaigns make a bigger impact when they explain the risks of DIY dosing and share the voices of real patients who learned the hard way.
Better Solutions for Users and Professionals
Open lines of communication between patients, clinicians, chemists, and even suppliers bring solutions to the table. Health providers should always offer clear directions about bismuth-containing treatments. Manufacturers should label packages truthfully and provide tested usage instructions. Regulatory agencies can increase random checks on supplement shelves and take quick action against gray-market sellers. For the curious or those tasked with handling bismuth carbonate, continuing education courses and up-to-date safety data sheets give real-world guidance that beats guesswork every time.